Diagnostic workflow for small intestine diverticulitis: case series and systematic review.
Objective: to assess new strategies in order to promptly diagnose small intestine diverticulitis.
Methods: we thoroughly analyzed a case series of small intestine diverticulitis in order to describe three of the typical presentations of such disease (intestinal perforation, intestinal obstruction and obstruction developing perforation). A systematic review of the literature was then conducted in five different scientific databases using six different keywords. Imaging technique's sensitivity for small intestine diverticulitis was assessed, as well as the frequency of signs and symptoms of such pathology. A complete description of the signs and symptoms of acute abdomen-type pathologies was also investigated in order to provide differential traits to distinguish small intestine diverticulitis.
Results: No pathognomonic signs or symptoms were detected for the pathology, although we found that all the other pathologies in the “acute abdomen” type of diseases have distinguishing traits that could be used to perform a differential diagnosis of small intestine diverticulitis. Moreover, contrast-enhanced computed tomography is the best imaging technique for the confirmation of such disease, when guided by an initial suspicion.
Conclusion: Small intestine diverticulitis cannot be diagnosed by clinical exploration only, but patients that have acute abdomen characteristics, and are negative to signs and symptoms that are characteristic of the other diseases in its type should be suspected for such disease. The aforementioned suspicion may guide a better imaging technique selection and exploration through it.
- We performed a thorough, systematic literature search finding that small intestine diverticulitis has no pathognomonic signs or symptoms.
- Contrast-enhanced computed tomography possess the highest sensitivity for its diagnosis when guided by suspicion of the pathology.
- Small intestine diverticulitis can be differentially diagnosed from other acute abdomen type pathologies.
Small intestine diverticulitis, diagnosis, differential diagnosis, acute abdomen.
*Correspondening autor: Alberto Navarrete-Peón, Laboratorio de Biomedicina Santiago Ramón y Cajal, Sociedad Española de Beneficencia, Av. Juárez #908, Pachuca, Hgo, PC 42060, 771 33 92 176, investigacion@benepachuca.com, alberto_navarrete@uaeh.edu.mx
- Introduction
A diverticulum is a blind tube or pouch that diverts from the gastrointestinal tissue, and can occur at any level, from esophagus to colon. Their most common localization for diverticula is the colon (50%), but they can also occur in the small intestine, which is rare as small intestine diverticulosis (SID) has a prevalence of 1-8% [1, 2]. Duodenal diverticulosis occurs in 6% of the population, whereas diverticula in the jejunum and ileum occur in 0.07-1% of the population [3]. Jejunoilel diverticula are more common in men, but duodenal diverticulosis occurs equally in both sexes [4].
Diverticula are classified according to their histological characteristics and etiologic origin in two main types: Meckel’s diverticula (MD), and non-Meckel’s diverticula (nMD). The former are thought to be an omphalomesenteric remnant that results from the failure of the vitelline duct to involute at the eight week of gestation, occasioning the outpouching of the lining mucosa, the submucosa, the muscularis, and the outer serosa [5]. On the other hand, the latter is thought to originate from the herniation of the mucosa, submucosa and serosa through the muscular layer at the blood vessel penetration sites [6] and are thought to be acquired after birth. Because of its morphology and origin, nMD diverticula are also known as false diverticula, but they have been described to be more prone to complications as their walls are thin and fragile [7]. However, MD tends to be asymptomatic except in 2-4% of the patients, with complication rates between 15-40% [8-10].
Although SID can course asymptomatically for 30-70 years, such abnormality may complicate itself to induce diverticulitis (2-6%), intestinal obstruction, intestinal perforation (2.1-7%), malabsorption, abscess, volvulus, generalized peritonitis, fistula and bleeding [5]. The mortality rate of such rare entity ranges from 0-5%, unless diverticular perforation occurs, which enhances the mortality rate to 40% [11]. On the other hand, 30-70% of the patients with SID present with additional diverticula in the colon, 2% in the esophagus, 2% in the stomach, and even 12% in the urinary bladder [12]. Additionally, 10-40% of the patients with jejunoiliel diverticulosis develop acute complications in their lifetimes [13, 14]. In this way, a delayed diagnosis is thought to be the main factor inducing a poor prognosis [15].
Nonetheless, diagnosis of this condition is challenging as all of its reported signs and symptoms (SS) are thought to be unspecific, which difficult differential diagnosis among varied conditions such as appendicitis, sigmoid diverticulitis, visceral perforation, malignancy, Crohn’s mass and intestinal duplication cyst, cholecystitis, and ovarian torsion or rupture [16]. Imaging studies have not been of much utility in the confirmation of SID diagnosis, as only 50% of symptomatic diverticulosis cases have been diagnosed through preoperative imaging [17]. In this way, diagnosis has been largely done by exploratory laparotomy, which is not ideal for its invasiveness.
The limited usefulness of imaging studies is thought to occur mainly because radiologists do not intentionally seek signs of SID, which, on its own, is a manifestation of a phenomenon where the gastroenterologists do not firstly suspect of this rare condition. Following this line of thought, Transue et al. (2016) have proposed that the imaging diagnosis of this entity should be based primarily on a clinical examination-derived suspicion of SID, and they think that multi-detector computed tomography (MDCT) with intravenous contrast should be the imaging choice for the confirmation of such entity [18].
We think that although no pathognomonic SS have been detected for this disease, an integral procedure designed to take into account the whole symptomatology to direct the search for SID on imaging may be useful to facilitate prompt diagnosis and thus aid in the avoidance of complications. Hence, hereby we present a case series with a systematic review of the literature that aims at enhancing the diagnostic ability of medical practitioners on such an uncommon entity.
- Materials and methods
Case series analysis. Our case series was firstly presented in a narrative form in order to summarize examples of two common SID presentations. Furthermore, all the SS that are commonly described to form part of an acute abdomen-type pathology were plotted into a table, and the SS that we found to be present on our cases were checked into such table (code: Y), whereas the SS that were not present in our patients were marked as absent (code: N). SS that are typical of acute abdomen-type pathologies, but were not recorded on the clinical files of each case were marked as not mentioned (code: 0).
Literature search. A systematic literature search for case reports of SID was made on PubMed, Embase, Web of Science, Cochrane Library and Science Direct, using the following keywords: small intestine diverticulitis, Meckel AND Non-Meckel diverticulitis, jejunal diverticulitis, duodenal diverticulitis, ileal diverticulitis, and jejuno ileal diverticulitis. Reference lists of relevant reviews were also screened for potentially eligible literature.
We utilized the following inclusion criteria: all the articles must be written in English, have included case reports of small intestine diverticulitis, a description of the signs and symptoms, and a diagnosis confirmation by histopathology or any endoscopic method giving a sound diagnosis. Moreover, the following exclusion criteria were used: articles older than 10 years at the moment of study, patients with gastrointestinal comorbidities, review and commentary articles that lack a case report or a case series, articles with no abstract. Duplicated articles and articles that mentioned asymptomatic cases were also eliminated.
Frequencies calculations. Based on the data from our case series, common SS of the pathology were detected and plotted into a table. The selected articles were read by three different researchers and the different SS that were reported were added to the table using the following nomenclature: Y, reported as positive; N, reported as negative; 0, not reported. The frequency of the different SS was calculated taking into account the sum of N and Y reports as 100%. 0-type reports were not taken into account as the lack of specific mention of the presence or absence of any SS does not allow for any inference about its occurrence.
- Results and disscusion
4.1 Case series
4.1.1 Case 1
A 69 years old patient presented himself to the hospital for type 2 diabetes and prostatic and arterial hypertension-related complications, nonetheless, 24 hr after his admission he started to present abdominal pain and distension, constipation, nausea, hematemesis, pallor, diaphoresis, abolished peristalsis and hyperbaralgesia, although he was Blumberg sign negative. Laboratory studies revealed high levels of amylase and seric urea, as well as leukocytosis with enhanced neutrophilia. An initial abdominal radiography showed severe ileitis with a thickening of the intestinal wall. After receiving antibiotics the gastrointestinal SS started to refer (48 hrs later), as he presented a higher intensity of peristalsis and a lower level of constipation. However, 11 days post admission he developed anemia, plus the initial gastrointestinal signs, and a computed tomography (CT)-scan showed thickening of the jejunal intestinal wall (Fig. 1e).
An exploratory laparotomy showed adhered jejunal handles surrounded by purulent material. A resection of the affected portion (40cm) was executed with posterior anastomosis of the remaining ends of the small bowel. The pathological study of the resected portion of the intestine showed multiple diverticula with chronic and acute diverticulitis (Fig. 1b).
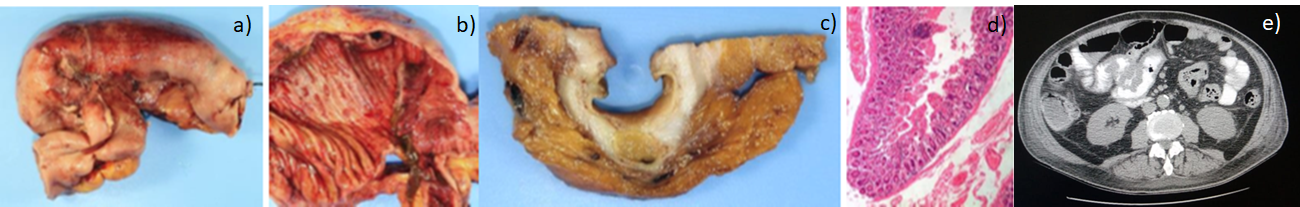
Figure 1. Small intestine diverticula. The recessed section of the jejunum (a), the opened recessed section (b) and the transversal cut of such section (c) show an evagination of the small intestine. The hematoxylin and eosin micrography of the evagination shows the morphology of a typical Meckel’s diverticulum (d). CT-scan showing the thickening of the intestinal wall (e).
4.1.2 Case 2
A 45 years old masculine patient presented to the hospital with diffuse abdominal pain that localized upon palpation of the right hypocondrium, nauseas, vomiting, dizziness, tachycardia, tachypnea, diaphoresis, dehydrated mucosal tissues, percussion tympanism, abdominal distention and resistance. Blood analysis revealed neutrophilia and high amylase levels, as well as anemia. On ultrasound (US) examination no SID was detected. Visceral perforation with septic shock of abdominal origin was suspected, so that an exploratory laparotomy was executed to find purulent secretions and a jejunal perforation.
The affected area, measuring 20cm, was fully resected to include the diverticula, which appeared as a blind pouch that neumatized when pressure was applied to the intestine walls. A complete anastomosis of the loose ends of the intestine was made and the pathological examination of the resected sample showed the presence of an MD (Fig. 2).
.png)
Figure 2. Jejunal diverticula. The recessed section of the jejunum (a), the opened recessed section (b) and the transversal cut of such section with hematoxylin and eosin staining (c) show an evagination of the small intestine consistent with the histopathological characteristics of an MD.
4.1.3 Case 3
A 79 years old male patient presented to the hospital with 8 days of diffuse abdominal pain that localized in the right hypocondrium upon palpation, abolished peristalsis, distended abdomen dehydration, astenia, adinamia, vomit and intense thirst. He also presented leukocytosis with neutrophilia.
CT-scanning was inconclusive, but showed extraluminal air. Exploratory laparotomy showed intestinal obstruction with an outpouching that was confirmed to be a perforated MD, so that the affected section was fully resected and the loose ends were anastomosed.
- Imaging approach to small intestine diverticulitis prompt diagnosis
As in our case series SID detection through imaging techniques was not possible we made a thorough systematic literature search for case reports of SID on PubMed, Embase, Web of Science, Cochrane Library and Science Direct, using a list of relevant terms to find case reports of SID with a description of the imaging techniques used to diagnose such pathology, and the findings of such studies with the aim of studying the sensitivity of the imaging studies. Seven articles were selected based upon diagnosis confirmation by histopathology or endoscopy studies giving a sound diagnosis (Figure 3).
Radiographies (XR) were used in three cases only, two of which reported no pathologic findings, and one of them reporting a gas artifact. US was performed on three cases, of which one found an unspecific obstructive pattern of the small intestine, and two reported a thickening of the intestinal wall that was surrounded by inflamed fat. CT was used for eight cases, one of which did not detected abnormalities, three of them were inconclusive, three found a thickening of the intestinal wall with surrounding inflamed fat and extraluminal air, and only one that reported a single diverticulum. Contrast-enhanced CT (CE-CT), on the other hand, was used in only three cases, with one describing multiple diverticula and two describing blind masses, either with air bubbles, or with enteroliths (Table 1). In this way, radiographies were rendered as poorly sensitive for SID (0% sensitivity), while US (66.6%), CT (66.6%), and CE-CT (100%) had better sensitivities (Fig. 4). On the other hand, Zhao et al. [19] report a similar sensitivity for small intestine endoscopy and CE-CT.
Nonetheless, although the findings of the imaging studies accurately describe the imaging signs of SID [18], the diagnosis is still made upon exploratory laparotomy as the interpretation of such findings is not commonly guided by a good clinical exploration. Moreover, not all imaging studies are highly sensitive for such pathology, so that prompt diagnosis may be delayed if a poorly sensitive study is performed to the patients. In this way we think that a more complete and precise clinical exploration may guide the imaging studies for enhanced sensitivity, which may aid in the prompt diagnosis of the illness.
| Case/Reference | C1 | C2 | C3 | [16] | [5] | [7] | [7] | [7] | [20] | [12] | [12] | [12] | [12] | [12] | [19] | [21] |
| SID signs detected | No | No | No | No | No | Yes | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Imaging study/findings | XR: severe ileum with edema; TAC: severe Ileum. | US: obstructive pattern. | CT: inconclusive. | CT: inconclusive. | CT and XR: normal. | CE-CT: multiple diverticula. | CT: Single diverticula. | n.a. | XR: gas artifact. | CE-CT: 5mm mass with air bubbles. | US: thickened wall with small projections. extra luminal fluid with air bubbles. Inflamed fat. | US: wall thickening surrounded by mesenteric fluid and inflamed fat. CT: wall thickening with decrease in pericentric fat. Extra luminal air. | CT: air and fluid collection surrounded by dilated jejunal loops with thickened walls and surrounding fat. | CT: thickening and hyper enhancement of the intestinal wall with fluid and air bubble collection. | CE-CT: 60% sensitivity. | CE-CT: blind ending sac with enterolithes, thickened wall with surrounding fat. |
Table 1. Imaging findings on small intestine diverticulitis.Abbreviations: C1/2/3: case 1/2/3; XR: radiography; US: ultrasound; n.a.: not available; CT: computed tomography; CE-CT: contrast-enhanced computed tomography.
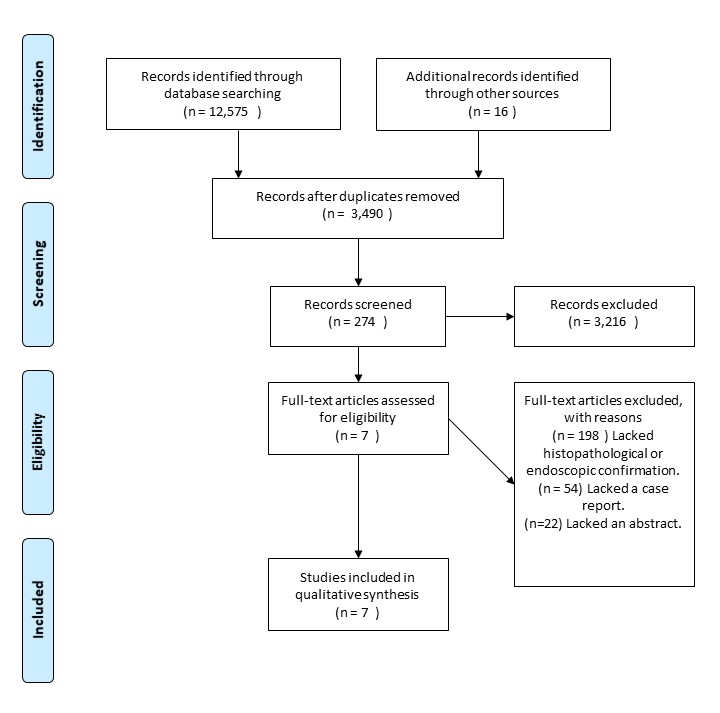
Figure 4: Sensitivity of the different imaging techniques for SID diagnosis. The sensitivity of the SS was calculated as percentage (%) of positive findings on confirmed cases of SID for each technique. Abbreviations: XR: radiography; US: ultrasound; CT: computed tomography; CE-CT: contrast-enhanced computed tomography.
4.3 Clinical presentation of small intestine diverticulitis
As mentioned above, SID is a challenging condition to diagnose, as no pathognomonic SS have been reported for the aforementioned entity, not all imaging studies are highly sensitive for such disease, and the interpretation of such studies is not guided by sound clinically acquired conclusions. In this way, we used our case series and the previously mentioned studies to extract data that may help to make a more complete description of the SS induced by this disease, and to calculate its frequencies.
Seven articles were selected to perform the calculation of SS frequency, in which we quantified a total of 21 variables that were selected from our case series (Table 2). The literature analysis showed that most articles reported few SS (present or absent) in comparison with those of our case series. In this way, we calculated SS frequency based on the articles that mentioned the presence (marked Y in table 2) or absence (marked N in table 2) of each variable, while taking out of the equation the reports that did not mentioned the presence or absence of a variable (marked as 0 in table 2), as its manifestation or lack of thereof cannot be inferred.
No pathognomonic SS were detected in our analysis, and the common presentation is that of acute abdomen [22] (Table 2), generally including abdominal pain syndrome (85.3%) that mainly manifests in the right ileac fossa or that is diffuse and localizes upon palpation in the mentioned area, digestive system bleeding (manifested as coffee grounds and blood in stools) (91.6%), constipation (75%), nausea (85.7%), vomiting (80%), abdominal resistance (83.3%), high seric urea (75%) and amylase (66.6%) levels, leukocytosis, fever, abdominal distention, dehydration, oedema, anemia, tachycardia, abolished peristalsis, pallor, hyperbaralgesia (100% for the aforementioned SS), and Blumberg sign (85%). Moreover, SID mostly occurs in men (76.4%), but it also has a high prevalence among women (23.5%), while the mean ages to present SID for both sexes are 71.1 for males and for females 62.8 (Table 3). However, from our case #1 we learnt that anemia and fever may only develop when a perforation occurs, while it may be absent in the setting of intestinal obstruction.
Table 2. Systematic review of the common SS of SID
| Variables/References | C1 | C2 | C3 | [1] | [2] | [3] | [3] | [3] | [4] | [5] | [5] | [5] | [5] | [5] | [6] | [7] |
| Sex | M | M | M | F | M | M | F | F | F | M | M | F | F | F | 18M 1F | M |
| Age | 69 | 45 | 79 | 45 | 75 | 85 | 86 | 72 | 20 | 82 | 75 | 48 | 83 | 86 | 15-60 | 59 |
| Abdominal pain síndrome | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | 5/19 | Y |
| Abdominal distention | Y | Y | Y | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Constipation | Y | Y | 0 | N | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Nausea | Y | Y | Y | 0 | 0 | N | Y | 0 | Y | 0 | 0 | 0 | 0 | 0 | 0 | Y |
| Vomiting | Y | Y | Y | 0 | 0 | N | Y | 0 | Y | Y | Y | 0 | Y | 0 | 0 | N |
| Pallor | Y | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Dehydration | Y | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Oedema | 0 | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2/19 | 0 |
| Abolished peristalsis | Y | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Hyperbaralgesia | Y | Y | Y | 0 | Y | 0 | 0 | 0 | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 |
| Blumberg | N | 0 | Y | 0 | 0 | 0 | 0 | 0 | Y | Y | 0 | Y | Y | 0 | 0 | Y |
| Fever | Y | Y | 0 | 0 | 38.5°C | 38°C | 0 | 0 | 37.6°C | 0 | 0 | 0 | 0 | 0 | 0 | 38.4°C |
| Tachycardia | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | Y |
| Anemia | Y | Y | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Leukocytosis | 11,000 cells/L | 15,580 cells/L | 17,500 cells/L | 0 | 18,100 cells/L | 13,800 cells/L | 11,400 cells/L | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 21,100 cells/L |
| Digestive system bleeding | Y | Y | 0 | N | Y | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 19 | 0 |
| Amylase | 205 U/L | 175.5 U/L | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | N |
| Seric Urea | 170 mg/dL | N | 269 mg/dL | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | Y |
| Abdominal resistance | N | Y | Y | 0 | Y | 0 | 0 | 0 | Y | Y | 0 | 0 | 0 | 0 | 0 | 0 |
Abbreviations: C1/2/3, case 1, 2 or 3; M, male; F, female; Y, reported as positive; N, reported as negative; 0, not mentioned in the report; U, units; L, liters.
Table 3. Frequency of SS for SID
| Criterion | Frequency |
| Age | Males: 71.1; females:62.8 (average) |
| Sex | 76.4% males, 23.5% females |
| Abdominal pain syndrome | 85.3% in the right ileac fossa |
| Digestive system bleeding | 91.6% |
| Constipation | 75% |
| Nausea | 85.7% |
| Vomiting | 80% |
| High amylase | 66.6%, mean: 190.25 U/L |
| Abdominal resistance | 83.3% |
| Leukocytosis | 100%, mean: 15,483 cells/L |
| Fever | 100%, mean: 38.1°C |
| Abdominal distention | 100% |
| Dehydration | 100% |
| Oedema | 100% |
| Blumberg sign | 85% |
| Anemia | 100% |
| Tachycardia | 100% |
| Abolished peristalsis | 100% |
| Pallor | 100% |
| Hyperbaralgesia | 100% |
Abbreviations: U, units; L, liters.
4.3 Differential diagnosis for SID
As the clinical presentation of SID possesses highly unspecific SS, that do not allow for a differential diagnosis among other acute abdomen pathologies, we next sought to determine if other SS that are absent from the typical SID presentation are, on the other hand, present in the other pathologies from the group.
In Table 4 we show that appendicitis, small intestine malignancy, ovaric cyst, perforated peptic ulcers and acute pancreatitis have four SS that render them as easily distinguishable from other acute-abdomen pathologies. Mesenteric cysts and cholelithiasis have three. Ovarian torsion, sigmoid diverticulitis and volvulus have two, and ruptured aortic aneurism has one that is, nonetheless, very distinctive. Crohn's disease is also easily recognizable because of different patterns of manifestations in eyes, mouth, skin and joints (such as uveitis and ankylosing spondylitis).
On the other hand, ovarian torsion and cysts occur exclusively in women, while SID is more common in men (71.1%). Also, mesenteric cysts and intestinal malignancies have a lower incidence than SID, which ranges at 1-8% [2].
Table 4. Differential diagnosis of SID.
| Diferential diagnosis | Cardinal signs | Incidence | Reference |
| Appendicitis | McBurney sign Talopercusion sign Rovsing sign Anorexia | 10/100,000 | [1] |
| Sigmoid diverticulitis | Lower left quadrant pain Transrectal bleeding | 5-65%, increasing with age (over 50% for 60 years old patients | [2] |
| Malignancy | Weight loss Change in intestinal habits Meleana Hematochezia Transrectal bleeding | 0.001-0.00227% | [3, 4] |
| Crohn's disease | Extraintestinal manifestations (in eyes, mouth, skin and joints) | 10.7 per 100,000 people | [5] |
| Cholelithiasis | Murphy’s sign Upper right quadrant pain and muscular resistance Mass in the upper right quadrant | 10-20% | [6] |
| Ovaric cyst | Dismenorrhaea Dispareunia Miccionary urgency or enhanced miccionary frequency | 7-21.1% | [7] |
| Mesenteric cyst | No haemetemesis No digestive system bleeding No pallor | 0.0004% of hospital admissions | [8] |
| Ovarian torsion | No digestive system bleeding Palpable mass in ileac fossas | 2.7% of gynecologic urgencies | [9] |
| Perforated peptic ulcer | Gastritis or Helicobacter infection history Peptic ulcer history Epigastric pain Dyspepsia | 1.5-3% of total population | [10] |
| Acute pancreatitis | Pain that migrates from the epigastrium to one or both sides Cooler sign Turner sign Fox sign | 4.9-80/100000 | [11] |
| Volvulus | Palpable mass Coffee bean, bird beak and whirl signs on CT | 2 per 100,000 individuals worldwide | [12] |
| Ruptured abdominal aortic aneurysm | Shock Hypotension Cold feet | 10 per 100,000 | [13] |
.png)
Figure 5. Algorithm for SID diagnosis. SID (small intestine diverticulitis), CE-CT (contrast enhanced computed tomography.
As a consequence, we think that a clinical exploration of the patients where an acute-abdomen pathology is found, but further exploration shows that none of the SS mentioned in Table 4 is present, must be suspected of being positive for SID, disregarding the low incidence of such pathology (Fig.5). In this way, the clinical exploration may guide a more precise interpretation of the findings of a highly sensitive imaging study (CE-CT).
- Conclusions
SID is a rare disease that possess no pathognomonic SS, and its clinical presentation is that of an acute abdomen pathology. However, we think that a complete exploration in which the cardinal SS of the other acute abdomen-type pathologies are discarded may provide a better clinical conclusion, in order to ask for a highly SID-sensitive imaging study and guide a more accurate interpretation of such diagnostic technique.
Such an enhanced diagnostic workflow may facilitate prompt diagnosis of SID, which may avoid further complications such as intestinal perforation and anemia, which greatly enhance the mortality of SID.
- Conflicts of interest
The authors declare that no potential conflicts of interest exist both on the writing and publication of this paper.
- Acknowledgements
All the authors wish to thank Sociedad Española de Beneficencia (Pachuca, Hidalgo) for funding the publication of this article. Moreover, Celia Nashielli Florez-Sánchez, Mario F. Gómez-Núñez and Kristopher Suárez-Bautista wish to thank the scholarship they receive from such Institution.
- References
1. Harbi, H., et al., Jejunal diverticulitis. Review and treatment algorithm. Presse Med, 2017. 46(12 Pt 1): p. 1139-1143.
2. Cunningham, S.C., C.J. Gannon, and L.M. Napolitano, Small-bowel diverticulosis. Am J Surg, 2005. 190(1): p. 37-8.
3. Miller, R.E., et al., Surgical complications of small bowel diverticula exclusive of Meckel's. Ann Surg, 1970. 171(2): p. 202-10.
4. Ferreira-Aparicio, F.E., et al., Diverticular disease of the small bowel. Case Rep Gastroenterol, 2012. 6(3): p. 668-76.
5. Syllaios, A., et al., Jejunal Diverticulitis Mimicking Small Bowel Perforation: Case Report and Review of the Literature. Chirurgia (Bucur), 2018. 113(4): p. 576-581.
6. Williams, R.A., et al., Surgical problems of diverticula of the small intestine. Surg Gynecol Obstet, 1981. 152(5): p. 621-6.
7. Ejaz, S., R. Vikram, and J.R. Stroehlein, Non-Meckel Small Intestine Diverticulitis. Case Rep Gastroenterol, 2017. 11(2): p. 462-472.
8. Gayer, G., et al., Acute diverticulitis of the small bowel: CT findings. Abdom Imaging, 1999. 24(5): p. 452-5.
9. Lee, N.K., et al., Complications of congenital and developmental abnormalities of the gastrointestinal tract in adolescents and adults: evaluation with multimodality imaging. Radiographics, 2010. 30(6): p. 1489-507.
10. Nigogosyan, M. and C. Dolinskas, CT demonstration of inflamed Meckel diverticulum. J Comput Assist Tomogr, 1990. 14(1): p. 140-2.
11. Kassir, R., et al., Jejuno-ileal diverticulitis: Etiopathogenicity, diagnosis and management. Int J Surg Case Rep, 2015. 10: p. 151-3.
12. Grana, L., et al., Jejuno-ileal diverticulitis with localized perforation: CT and US findings. Eur J Radiol, 2009. 71(2): p. 318-23.
13. Liu, C.Y., et al., Analysis of clinical manifestations of symptomatic acquired jejunoileal diverticular disease. World J Gastroenterol, 2005. 11(35): p. 5557-60.
14. Akhrass, R., et al., Small-bowel diverticulosis: perceptions and reality. J Am Coll Surg, 1997. 184(4): p. 383-8.
15. Krishnamurthy, S., et al., Jejunal diverticulosis. A heterogenous disorder caused by a variety of abnormalities of smooth muscle or myenteric plexus. Gastroenterology, 1983. 85(3): p. 538-47.
16. Abbas, S.H., et al., Mesenteric Meckel's diverticulum: an unusual cause of small bowel intussusception. BMJ Case Rep, 2016. 2016.
17. Kouraklis, G., et al., Clinical implications of small bowel diverticula. Isr Med Assoc J, 2002. 4(6): p. 431-3.
18. Transue, D.L., et al., Small bowel diverticulitis: an imaging review of an uncommon entity. Emerg Radiol, 2017. 24(2): p. 195-205.
19. Zhao, L., et al., Small intestinal diverticulum with bleeding: Case report and literature review. Medicine (Baltimore), 2018. 97(9): p. e9871.
20. Emre, A., et al., Double Meckel's diverticulum presenting as acute appendicitis: a case report and literature review. J Emerg Med, 2013. 44(4): p. e321-4.
21. Wong, C.S., et al., Meckel's diverticulitis: a rare entity of Meckel's diverticulum. J Surg Case Rep, 2017. 2017(1).
22. Mazzei, M.A., et al., The role of US examination in the management of acute abdomen. Crit Ultrasound J, 2013. 5 Suppl 1: p. S6.
23. Ferris, M., et al., The Global Incidence of Appendicitis: A Systematic Review of Population-based Studies. Ann Surg, 2017. 266(2): p. 237-241.
24. Weizman, A.V. and G.C. Nguyen, Diverticular disease: epidemiology and management. Can J Gastroenterol, 2011. 25(7): p. 385-9.
25. Pan, S.Y. and H. Morrison, Epidemiology of cancer of the small intestine. World J Gastrointest Oncol, 2011. 3(3): p. 33-42.
26. Aparicio, T., et al., Small bowel adenocarcinoma: epidemiology, risk factors, diagnosis and treatment. Dig Liver Dis, 2014. 46(2): p. 97-104.
27. Lichtenstein, G.R., et al., ACG Clinical Guideline: Management of Crohn's Disease in Adults. Am J Gastroenterol, 2018. 113(4): p. 481-517.
28. Stinton, L.M. and E.A. Shaffer, Epidemiology of gallbladder disease: cholelithiasis and cancer. Gut Liver, 2012. 6(2): p. 172-87.
29. Farghaly, S.A., Current diagnosis and management of ovarian cysts. Clin Exp Obstet Gynecol, 2014. 41(6): p. 609-12.
30. Pithawa, A.K., A.S. Bansal, and S.P. Kochar, "Mesenteric cyst: A rare intra-abdominal tumour". Med J Armed Forces India, 2014. 70(1): p. 79-82.
31. Huang, C., M.K. Hong, and D.C. Ding, A review of ovary torsion. Ci Ji Yi Xue Za Zhi, 2017. 29(3): p. 143-147.
32. Chung, K.T. and V.G. Shelat, Perforated peptic ulcer - an update. World J Gastrointest Surg, 2017. 9(1): p. 1-12.
33. Humes, D.J. and J. Simpson, Acute appendicitis. BMJ, 2006. 333(7567): p. 530-4.
34. Zabeirou, A.A., et al., Acute cecal volvulus: A diagnostic and therapeutic challenge in emergency: A case report. Ann Med Surg (Lond), 2019. 48: p. 69-72.
35. Schmitz-Rixen, T., et al., Ruptured abdominal aortic aneurysm-epidemiology, predisposing factors, and biology. Langenbecks Arch Surg, 2016. 401(3): p. 275-88.
Widget de imagen
Widget de imagen
Widget de imagen
Widget de imagen
Widget de imagen

 0
0
 0
0