The five axes of COVID-19 treatment: attacking the disease from several angles.
The emergency situation of the COVID-19 pandemics requires immediate action. As happens with emerging pathogens, there are no specific treatments for this threat, so that the most logical answer in order to find safe and effective candidates seems to be drug repurposing. The main efforts in finding a specific treatment for this disease have been directed to finding antiviral agents, nonetheless, COVID-19 also involves lung and systemic inflammation, coupled with ineffective immunity; bacterial and fungal coinfections; respiratory dysfunction; and coagulopathy. These additional pathophysiologic axes also require a set of treatments, and in this review we will analyze such adjunctive therapies.
The main pathophysiological mechanisms of COVID-19 are described.
Several strategies to cope with the pathophysiologic traits of the disease are described.
COVID-19; SARS-CoV-2; antiviral; antinflammatory; antithrombotic; antimicrobial; respiratory support.
- Introduction
Seven months after its appearance, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative agent of coronavirus disease (COVID-19), has caused more than 13 million infections and more than 500,000 deaths worldwide (1), also producing a severe economic impact throughout the world (2). The most common clinical manifestations of COVID-19 include fever, dry cough, fatigue, while less common symptoms include sore throat, headache, conjunctivitis, loss of taste and smell, skin rash or toe and finger discoloration and a small population of patients appeared gastrointestinal infection symptoms (3), and shortness of breath, chest pain and loss of speech and movement pinpoint a severe manifestation. Current research has shown an incubation period of 1-14 days, usually ranging from 3-7 days (4). It is highly transmissible among humans by droplets and contact with contaminated surfaces, and it is especially severe in the elderly and people with underlying diseases. The median age of patients is 47-59 years, and 41.9-45.7% of patients are females (5).
As such disease recently emerged, there is not known specific treatment to aid patients in their recovery, or even to increase survivability, but the rapid spread of SARS-CoV-2 has generated the urgent need to develop novel pharmacological therapies either by repurposing drugs that are usually prescribed to fight against other similar acute respiratory disease-inducing viruses, like SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV), or by developing novel specific therapeutic alternatives to fight this new pandemic. Nonetheless, the emergency of the situation calls for drugs that may be readily available, so that drug repurposing trials are currently being performed to assess the efficacy of currently existing and optimally widespread drugs (6), like lopinavir alone (7) or in combination with ritonavir, umifenovir (8), favipiravir (9), hydroxycholoquine and azithromycin combinations (10, 11), ivermectin (12), remdesivir (13, 14), tocilizumab (15, 16), heparin (17) as well as other antiplatlet agents, dexamethasone (18) and other anti-inflammatory, immunomodulating and antibiotic drugs.
As COVID-19 pathophysiological cornerstones include not only the viral causal agent, but also lung and systemic inflammation, lymphopenia, bacterial respiratory co-infections, respiratory disfunction, thrombocytopaenia and coagulopathy (19-21), we think that the treatment regimens combining antiviral, antinflammatory and antiplatelet agents are going to provide an enhanced clinical efficacy over monotherapies. Thus, in the present article we will review the most interesting proposals for anti-COVID-19 chemotherapy, emphasizing in drug combinations to attack the disease.
- The five axes of COVID-19 pathophysiology
SARS-CoV-2 is a linear positive sense, single-stranded RNA virus, thus belonging to the Baltimore group IV of viruses. For such virus type, RNA alone is infectious as it can be translated by the ribosome to synthesize new virions, while the virus’s genome encodes an RNA-dependent RNA polymerase (RNA replicase or RdRp) that generates copies of the genome within the host cell (22). Recent research by Hoffmann and colleagues (23) shows that this virus is able to interact with the host cell membrane by the binding of the viral spike (S) proteins with the host´s transmembrane protease serine 2 (TMPRSS2) to prime the S protein and with the angiotensin-converting enzyme 2 (ACE2) to penetrate the host’s cell. ACE2 is highly expressed in the small intestine, lungs, heart, and adipose tissue (24, 25). On the other hand, TMPRSS2 expression is high in bronchial transient secretory cells (26), thus leading to an enhanced lung tropism. Moreover, ACE2 has been recently described as an interferon-stimulated gene (27, 28), which may signify that SARS-CoV-2 is able to enhance its life cycle through the induction of inflammation (Fig. 1).
COVID-19, on the other hand, ranges from mild symptoms to severe respiratory and multiple organ failure. Typically, the virus enters the lung epithelial cells (pneumocytes) in the alveolar space and depletes them (29, 30), thus producing pulmonary edema, diffuse alveolar injury with cellular fibromyoxide exudates, hyaline membrane formation, pneumocyte hyperplasia, proteinaceous aggregates, fibrinous exudates, multinucleated syncytial cell formation and inflammatory infiltration of interstitial mononuclear cells (31-33), bronchitis and the characteristic pulmonary ground glass opacifications that can be seen even in the computed tomographies (CT) of asymptomatic patients (34). About 15% of the total patients will develop severe pneumonia, while 17-29% of the hospitalized patients will develop acute respiratory distress syndrome (ARDS) upon lung damage accumulation (35, 36). As such, enhanced levels of C-reactive protein (CRP), lactic dehydrogenase (LDH), ferritin, D-dimer, and IL-6 represent common findings on the laboratory examination of COVID-19 patients (Fig. 1).
COVID-19-associated ARDS results from a systemic inflammatory response with the subsequent appearance of diffuse alveolar damage accompanied by epithelial cell destruction (37). In addition to being the entry channel for the virus, it has been described that ACE2 physiologically degrades angiotensin II, which promotes inflammation and vasoconstriction, to convert it into angiotensin 1 or 7, which are normally involved in lung protection. Therefore, it has been stipulated that in SARS-CoV-2 infections an antagonism of ACE2 occurs, which may in turn promote lung damage (38) (Fig. 1).
On the other hand, damage-associated molecular patterns (DAMPS) and pathogen-associated molecular patterns (PAMPS) produced during the viral damage, or by the pathogen itself have been implicated in the M1 activation of macrophages (39) and monocytes (40) through Toll-like receptor signaling, NLRP3 inflammasome activation and triggering of cytoplasmic DNA sensors. In such an activation state, these cells contribute to the lung epithelial damage that leads to ARDS, while their production of IL-6 and IL-1β recruit neutrophils and cytotoxic T cells. Within the lung parenchyma, neutrophils produce vast amounts of reactive oxygen species (ROS) and leukotrienes that potentiate both the antiviral response and the immunopathology. In fact, IL-6 levels have been associated with a poor prognosis for the patients (41).
Such observations have led to the hypothesis that COVID-19 patients develop a “cytokine storm” (42), which is not only based on the aforementioned cytokines, but also includes IL-2, IL-2R, IL-7, IP10, MIP1A, TNF-a, IFN-γ, IP-10, MCP-1 and granulocyte-colony stimulating factor (G-CSF) (43, 44), but strikingly, is also complemented by Th2-type cytokines like IL-10 and IL-4 (44). While patients do appear to die primarily after respiratory failure, but not after the aforementioned cytokine storm, it cannot be inferred that the main mechanism for COVID-19 lethality is excessive inflammation, however such immunologic traits should be taken into account as they may offer clues to find the correct pathways to treat COVID-19, or to predict its outcome. For instance, the severe manifestation of this disease is manifested by higher levels of IL-1β, IL-2R, IL-6, IL-8, IL-10, and TNF-a than those presented by the asymptomatic or moderately ill patients (41, 45).
Furthermore, viral dissemination appears as a cornerstone for COVID-19 complications, and the production of type I and III interferons appears to be critical in the control of such phenomena (39), but effective clearance of the viral load requires CD8+ and CD4+ T-cell responses as well as B-cells, as such cells appear to be the main sources of these cytokines. Nonetheless, COVID-19 patients usually present with low levels of these cytokines (46) and their total numbers of CD8+ and CD4+ T-cells are markedly reduced in severely ill patients, while the expression of high levels of the coinhibitor signaling receptors programmed death-1 (PD-1) and Tim-3 are overexpressed in such cells, especially in those patients that presented enhanced IL-6, IL-10, and TNF-α concentrations (47) (Fig. 1). Furthermore, a negative correlation between serum levels of IL-6 and IL-8 and the perforin content of NK and CD8+ cells has been described, while high concentrations of these cytokines also correlate with an increased number of circulating myeloid-derived suppressor cells (MDSC) (45), and lower levels of CD8+ cells also predict worse outcomes (45).
While it has been hypothesized that antibodies against SARS-CoV-2 may be able to block the S1-ACE2 interaction, thus avoiding viral penetration, and IgM and IgA antibodies can be found during the first week of symptom onset and IgG can be found after 14 days of such event (48), evidence pointing at a significant decay of immunity to SARS-CoV after six years suggests that the immunity against coronaviruses may be not long lasting (49). Additionally, in severe COVID-19, the number of circulating naive T cells increases while the number of memory T cells decreases (50).
On the other hand, COVID-19 patients present with intense coagulopathy, in such a way that elevated D-dimer levels (higher than 1 μg/ml) and disseminated intravascular coagulation at the time of hospital admission have been strongly correlated with an increased mortality (51). Such hypercoagulative state in COVID-19 patients is thought to be mainly derived from the activation of the inflammatory response and CRP production, which increases the exposure to Tissue Factor in monocytes and alveolar macrophages, and in turn promotes thrombin generation and fibrin deposition (52). In such circumstances, IL-6 is a main inductor of coagulopathy, as it induces the production of CRP in the liver. Furthermore, IL-1 and TNF-α synergize with IL-6 to activate the coagulation system. Taking into account that both inflammation and coagulation are two of the most important immune defense systems, this bidirectional communication of inflammation and coagulation is known as immunothrombosis (53). Coagulopathy associated with COVID-19 may occur in wide spectrum, including sepsis-induced coagulopathy and disseminated intravascular coagulation, hemophagocytic syndrome, antiphospholipid syndrome, and thrombotic microangiopathy (54) (Fig. 1).
Coagulopathy does not only occur systemically, as the aforementioned cytokines activate the coagulation process within the alveolar space, allowing fibrosis without progressing to fibrinolysis, which explains the elevated levels of d-Dimer. Some substrates, such as polyphosphates, activated platelets, mast cells, and the coagulation factor XII contribute to the activation of clotting factors, while neutrophil extracellular traps (NETs) help the formation of thrombin, and PAMPs/DAMPs maintain the interaction between the immune response and coagulation (53).
Such crosstalk between the thrombotic and inflammatory mechanisms, along with the adhesion of SARS-CoV-2 to the endothelial barrier through its interaction with ACE2, is thought to produce endotheliopathy by inducing apoptosis of the endothelial cells, and this phenomenon may enhance localized coagulopathy (51, 53). Furthermore, endotheliopathy could explain cerebrovascular complications in patients of early age, as well as the significant increase in macrocirculatory and microcirculatory thromboembolic complications in patients with this disease.
Finally, as lymphopenia occurs, causing an immune compromise, one of the issues involved in this disease is the induction of bacterial or fungal respiratory coinfections, which ranges from 7 to 94% of the patients (55-57). Although some studies suggest that these pathogens can contribute to the severity of respiratory pathology, there is no evidence of any significant difference in this regard. The most common co-infecting pathogens are Streptococcus pneumoniae, Klebsiella pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, and Aspergillus(55) (Fig. 1).
In such panorama, uncontrolled viral proliferation and dissemination appears to be a consequence of an impaired adaptive immune response, while an enhanced innate immune response may be responsible for enhanced inflammation and immunopathology, leading to ARDS (58). In this way, the stimulation of adaptive immunity and the simultaneous downmodulation of the innate immune response may be good therapeutic alternatives to manage the viral load and the pathologic inflammation produced in COVID-19. In concrete, the neutralization of key cytokines like IL-1β, IL-6, and IL-10, and the blockade of PD-1 may represent good alternatives to boost the anti-SARS-CoV-2 immune response.
Furthermore, IL-6 neutralization may be a staple in COVID-19 treatment as it may also reduce coagulopathy, while PD-1 blockage or any other form of immunostimulation may help to reduce the incidence of respiratory coinfections, but this issues should be treated diligently as they appear to be cornerstones of COVID-19 pathology.
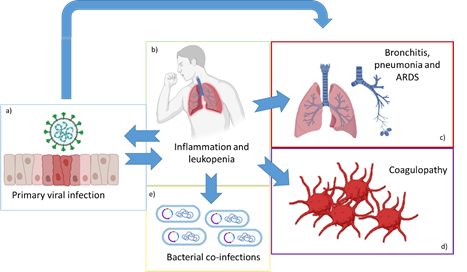
Figure 1. The five axes of COVID-19 pathology. The viral infection (a) produces lung tissue damage (c) as well as inflammation and leukopenia (b), which in turn enhances lung damage and ARDS (c), coagulopathy (d), bacterial co-infections (e) and may enhance viral penetration through the induction of ACE2 by IFN-γ production.
- Repurposed drugs against SARS-CoV-2 and COVID-19: attacking the disease from several angles
As eight months after its appearance, SARS-CoV-2 has caused more than 13 million infections and more than 500,000 deaths worldwide (1), this pandemics represents a worldwide sudden emergency that requires immediate action. In this way, the discovery of new specific drugs to fight this infection is not a viable option, as such a strategy would require more than a decade to produce results (59). A more viable option to combat such a fast-spreading infection is the repurposing of existing drugs, as they are mass produced, widely available, approved by sanitary-controlling organizations in many countries, their posology is well known and there is enough data about their safety (60).
Nonetheless, the current pharmacopeia is underexploited, when there is much potential to fight even emerging diseases as COVID-19. We think that drugs that possess anti-SARS-CoV-2 actions (being antiparasitic, antipaludic, or straight antiviral drugs) have been gathering much attention, when in reality COVID-19 is a more complex disease where coagulopathy, co-infection, inflammation and the lack of an effective immune response, as well as respiratory support must also be addressed (Fig. 2). In such an understanding, we think that an exhaustive review covering the varied awns of COVID-19 pathophysiology and drugs that may help to treat them may be of importance for the sake of treating the disease effectively.
.png)
Figure 2. The five axes of COVID-19 treatment: support for the acute respiratory distress syndrome may benefit from positively ionized oxygen and prone positioning of the patient; while immunopathology and inflammation may be regulated through key cytokine antagonists, corticosteroids and immune-stimulating agents; coagulopathy may be addressed with low molecular weight heparins; co-infections have been controlled through macrolide, quinolones and beta-lactams; and many straight antiviral of repurposed anti-viral drugs are currently in investigation.
- The antiviral axis
Many excellent research that is based on in vitro, in vivo and/or on experience with several Baltimore IV-type viruses like Zika virus, SARS-CoV, and MERS-CoV; or even with viruses that belong to other Baltimore types, but share any life-cycle-related traits with SARS-CoV-2; suggest a group of drugs that are aimed to target the SARS-CoV-2 viral replication cycle, like inhibiting the fusion between the S protein and the host’s ACE2 and TMPRSS2, translation and proteolysis by the 3-chymotrypsin-like protease, replication mediated by RdRp enzyme, inhibition of the heterodimer IMPα/β1-mediated nuclear import, or by modifying host’s cytological traits like the endosomal pH (Fig 1).
3.1.1 Remdesivir
Remdesivir is a phosphoramidite prodrug of adenosine C-nucleoside with a broad-spectrum antiviral activity. The active form of remdesivir (GS-441524) has to reach the inside of the cell to undergo rapid conversion to the pharmacologically active metabolite form GS-443902, which is a nucleoside triphosphate that acts as an analogue of natural ATP and binds the nascent RNA chains of the RdRp enzyme, thus producing premature RNA chain termination (61, 62). Such mechanism of action renders Remdesivir as a good alternative to treat Baltimore type IV viruses, and has a history of being useful in the experimental inhibition of the SARS-CoV and MERS-CoV life cycles, which rendered it as a good alternative to assay against SARS-CoV-2 (63) (Fig. 3).
Recent investigations by Grein et al (64) show that 53 Remdesivir-treated patients presented a clinical improvement in 68% of the cases, as measured by oxygen support class, including a 57% of patients that were extubated. Only 13% of the treated patients died, which is a good number considering that all of them were critically ill during the study. Nonetheless, a bigger (237 patients) randomized, double-blind, placebo-controlled, multicentre trial by Wang and colleagues (13) found that this drug does not produce a statistically significant clinical improvement, but recognized the need of a more powerful study to tell if such difference exists. Such study came from the team of Beigel and colleagues (14), who conducted a double-blind, randomized, placebo-controlled trial including 1059 patients that showed a reduction of ≈4 days of hospital stay and nearly a 5% reduction of mortality on the Remdesivir-treated group (Table 1).
3.1.2 Lopinavir/Ritonavir
Lopinavir is a viral protease inhibitor while Ritonavir is a potent inhibitor of the enzymes that are responsible for lopinavir metabolism (CYP3A4). The low oral bioavailability and extensive biotransformation of lopinavir make necessary the co-administration of lopinavir with ritonavir to act as a pharmacokinetic enhancer (65). Lopinavir by itself is a peptidomimetic molecule which prevents the activity of viral 3-chymotrypsin-like protease (3CLpro) by a hydroxyethylene scaffold that mimics the peptide linkage typically targeted by such viral protease, but cannot be cleaved, thus occupying the active site of the enzyme and inhibiting it (66). This drug combination has been essential to treat HIV infections (Fig. 3).
Like HIV, SARS-CoV-2 expresses the 3CLpro enzyme to control its replication, thus being an essential enzyme for these viruses life cycle (67), and the fact that the Lopinavir/Ritonavir drug combination has shown to inhibit this coronavirus enzyme with enhanced affinity (68) pinpoints at a possible role for this drug in the treatment of COVID-19.
A small clinical trial including 47 patients showed a reduced body temperature, C-reactive protein (CRP) levels, alanine aminotransferase and aspartate aminotransferase as well as enhanced lymphocyte numbers that correlated with a quicker reduction of the viral load (69) in relation to the Lopinavir/Ritonavir treatment, while other authors found that this drug combination was also able to increase the level of oxygen saturation and pressure, platelet, lymphocyte, leukocyte and eosinophil counts, while reducing the extension of radiological findings (7). Moreover, while a bigger study including 199 patients showed no reduction in viral load in relation to the Lopinavir/Ritonavir drug combination, they found that mortality decreased 5.8% in the treated group (70). Finally, another trial showed that a combination of interferon β-1b, Lopinavir-Ritonavir, and ribavirin works significantly better than Lopinavir/Ritonavir alone in alleviating symptoms, shortening the duration of viral shedding, and reducing hospital stay in patients with mild to moderate COVID-19 (71). Moreover, a study of 101 patients receiving different combinations of Ribavirin, Interferon-a and Lopinavir/Ritonavir, showed that no treatment was able to reduce the viral load or shorten the viral shedding phase in the patients. Instead, the patients receiving Ribavirin plus Lopinavir/Ritonavir presented a high prevalence of gastrointestinal adverse effects (72) (Table 1).
3.1.3 Umifenovir
Arbidol, the brand name of Umifenovir, is a broad spectrum antiviral drug that has reported activity against A and B type influenza and other arboviruses (73) , hence the name, but it also has in vitro antiviral activity against Ebola virus, human herpesvirus 8, hepatitis C virus and Tacaribe arenavirus (74). This drug has been reported to block viral fusion with the host cell membrane and the endosomes by interfereing with the normal structure of phospholipids (75). More importantly, Arbidol has been shown to impede the trimerization of the SARS-CoV-2 S protein (76), a vital step for the viral cycle of such pathogen (Figure 3).
Although a retrospective study in a non-intensive care unit made by Lian and colleagues showed that Umifenovir is not able to accelerate clearance of the viral load by day 7 post-treatment onset (PTO) (77), a similar study by Zhu et al. (8) showed that it lowers such parameter in a delayed manner, as measured 14 days after hospital admission, being superior to Lopinavir/Ritonavir in that regard. Aditionally, a metanalisis performed by Siordia’s team (78) showed no 7-day or 14-day viral clearance difference compared with the aforementioned drug combination. Finally, when given as prophylactic to health-care professionals, Arbidol was able to reduce the incidence of new cases, although it had no effect in reducing the hospitalization rate of those who became infected (79) (Table 1).
3.1.4 Favipiravir
Favipiravir triphosphate is a purine nucleoside analogue, which acts as a competitive inhibitor of RNA-dependent RNA polymerase (80), therefore inhibiting the in vitro replication of a wide array of viral infections, including Ebola virus (81), oseltamivir and zanamivir-resistant influenza virus strains (82) (Fig. 3). Although its in vitro effect against SARS-CoV-2 virus has been assayed successfully, both Chloroquine and Remdesivir outperform this drug at lower doses (83). Nonetheless, a small clinical trial of 80 patients with mild or moderate COVID-19 found that this drug produced a faster viral clearance (7 days less) than Lopinavir/Ritonavir and a considerable improvement in chest imaging (91.43% versus 62.22%) (9). Therefore, more data is needed in order to confirm whether or not Favipiravir may be a good and valuable option for the treatment of such viral infection.
3.1.5 Ivermectin
Ivermectin is an antiparasitary drug that is able to reduce the in vitro replication of some Baltimore type IV viruses, like Dengue virus (84), Zika virus (85) and yellow fever virus (86), that is generally considered as safe and effective for its original prescription. As this drug has been identified as an inhibitor of importin α/β-mediated nuclear transport of viral products (84) (Fig. 3) and a recent in vitro study showed that this drug is able to exert a ≈5000-fold reduction of the viral RNA after 48 hours or its addition into a SARS-CoV-2-infected cell line (12), the scientific community gained interest in this drug as a potential treatment for the infection with SARS-CoV-2. Nonetheless, such study was carried out using a dose that is 10 times higher than the one approved by the FDA (87), and the Pan American Health Organization declared that the studies about the use of such drug to control SARS-CoV-2 are strongly biased (88), in such a fashion that the World Health Organization (WHO) retired this drug from the Solidarity trial (89) for the lack of scientific evidence about its efficacy in the treatment of COVID-19.
Well designed and unbiased studies about this drug are needed in order to confirm its hypothetical effect on the control of this emerging disease (Table 1).
3.1.6 Chloroquine/Hydroxychloroquine
Chloroquine, and its less toxic hydroxylated form, are anti-malarial drugs (90) that have been repurposed for its use against SARS-CoV-2. Moreover, this drug has been shown to deter the in vitro virus-host cell fusion by interfering with the glycosylation of the ACE2 protein (91-93). Moreover, after penetration the virus releases its genome and some enzymes into the cytoplasm by fusing with the lysosomal membrane, in a process aided by the acidic pH of the aforementioned cell structure (Fig. 3). In concordance, chloroquine has been able to impede this process in other enveloped-virus dependent infections, like Chikungunya and Dengue, in association with the alkalization of the lysosome (94). Further evidence described that this drug is also able to reduce the levels of IL-6 in individuals with systemic lupus erythematosus and rheumatoid arthritis (95), thus hypothetically reducing the chance to develop cytokine storm and ARDS-related complications (96).
On the other hand, a study by Gautret et al. (97) that included 36 patients reported that Choloquine is able to significantly reduce the viral load at day 6 PTO, while the patients that also received Azithromycin had the fastest viral clearance. And a further study made by the same team (98) including 80 patients reported that at day 7 PTO 83% of the patients were negative for viral load and 93 were completely free of the virus by day 8. However, these two articles were criticized for their low power and the lack of other measured outcomes. In such an understanding, Million and colleagues (99) performed a much more powerful study including 1061 patients that were treated with a Chloroquine and Azithromycin combination, observing a virological clearance for 91.7% of the patients at day 10 PTO, and a 100% were cleared at day 15 PTO. 4.3% of the patients evolved to a poor clinical outcome and only 0.75% died. Nonetheless, Geleris and his team (100) found no differences in the mortality or intubation-risk of the hydroxychloroquine group. Despite the increase on the studies power, none of the aforementioned studies included severely ill patients, which only served to increase the controversy on this drug´s use.
As a consequence, Mahévas et al. (101) performed a comparative analysis of 181 patients with a severe form of COVID-19 that required oxygen supplementation but not intensive care, finding no difference in the survival rate without transfer to the intensive care unit at day 21, overall survival, ARDS incidence or changes in respiratory support requirements between the hydroxychloroquine and the standard care group, thus suggesting that this drug do not have clinically measurable benefits for the treatment of this disease in patients with severe disease. Finally, Sevilla-Castillo et al. (102) performed a study with severely-ill patients, where Chloroquine was used in combination with Lopinavir/Ritonavir and found that the drug was unable to modulate the pathology, but rather disease markers increased up to ≈13 fold when both drugs were combined (Table 1).
Table 1. Anti-SARS-CoV-2 drugs.
| Drug | Mechanisms of action | Evidence | References |
| Lopinavir/Ritonavir | Viral 3-chymotrypsin-like protease (3CLpro) inhibitor. | Reduced temperature, CRP, ALT and AST. Enhanced lymphocyte and viral clearance time. (n=47) Enhanced oxygen saturation and pressure, platelet, lymphocyte and eosinophil counts. Reduced radiological findings. (n=10) No reduction in viral load, 5.8% decrease in mortality. (n=199) Combination of interferon β-1b, Lopinavir-Ritonavir, and ribavirin alleviate symptoms, shortens the duration of viral shedding, and reduced hospital stay in patients with mild to moderate COVID-19. (n=127) No reduction of the viral shedding phase, plus enhanced gastrointestinal adverse reactions. (n=101) No reduction of mortality, hospitalization, respiratory requirements, CRP and LDH; significant increase in d-dimer and ferritin. | (69) (7) (70) (71) (72) (102) |
| Ivermectin | Inhibitor of importin α/β-mediated nuclear transport of viral products. | ≈5000-fold reduction of the viral RNA after 48 hours of its addition into a SARS-CoV-2-infected cell line. The working dose is 10 times higher than recommended. | (12) |
| Chloroquine | Interferes with the glycosylation of the ACE2 protein and alkalizes the lysosome. Reduces IL-6 levels. | Reduces the viral load at day 6 PTO. (n=36) At day 7 PTO 83% of the patients are free of viral load and 93% were completely free of the virus by day 8. (n=80) Virological clearance for 91.7% of the patients at day 10 PTO, and a 100% were cleared at day 15 PTO. 4.3% of the patients evolved to a poor clinical outcome and only 0.75% died. (n=1061) No differences in the mortality or intubation-risk of the hydroxychloroquine group. (n=1446) No difference in the survival rate without transfer to the intensive care unit at day 21, overall survival, ARDS incidence or respiratory support requirements. (n=181) | (97) (98) (99) (100) (101) |
| Favipiravir | Competitive inhibitor of RNA-dependent RNA polymerase. | Faster viral clearance then lopinavir/ ritonavir and improved chest imaging 91.43 % of the patients. (n=80) | (9) |
| Umifenovir | Blocks viral fusion with the host cell membrane and the endosomes. | No reduction of the viral load at day 7. (n=81) Reduction of the viral load at day 14, being superior to Lopinavir/Ritonavir. (n=50) A study showed no 7-day or 14-day viral clearance difference in comparison to the aforementioned drug combination. As a prophylactic to health-care professionals, it was able to reduce the incidence of new cases, although it did not have any effect in reducing the hospitalization rate of those who became infected. (n=164) | (77) (8) (78) (79) |
| Remdesivir | Inhibitor of the RdRp enzyme | Clinical improvement in 68% of critically ill patients, as measured by oxygen support class. (n=53) Does not produce a statistically significant clinical improvement. (n=237) Reduction of ≈4 days of hospital stay and a 5% reduction of mortality. (n=1059) | (64) (13) (14) |
Abbreviations: C-reactive protein (CRP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), Interleukin-6 (IL-6), post-treatment onset (PTO), acute respiratory distress syndrome (ARDS).
.png)
Figure 3. Mechanisms of action of the anti-SARS-CoV-2 drugs.
3.2 The immunoregulatory axis
As the virus-induced inflammatory response is thought to contribute significantly to the development of the whole pathology, a paramount of its pharmacological control is the modulation of inflammation. As such, corticosteroid administration has been considered to be a good option for COVID-19 adjunctive therapy. To date, preliminary results from the RECOVERY trial show a reduction of 12.1% on the 28-day mortality of patients receiving invasive mechanical ventilation in relation to dexamethasone administration, but an increase of 3.8% of such parameter when this drug was administered to patients that were not receiving respiratory support (103). In a similar fashion hydrocortisone and methylprednisolone have also been associated with enhanced 28-day survival (104). On the other hand, Li et al. (105) observed a prolonged duration of fever, viral shedding stage and length of hospitalization, as well as an enhanced need for antibiotics in patients that received corticosteroids.
In such panorama, there are important gaps in the knowledge regarding the use of such drugs in the treatment of COVID-19, and answering some key questions may be critical for the optimization of such drug-class use. For instance, being that corticosteroids are immunosuppressant agents, they may favor viral shedding if their administration occurs at the wrong time or dose, and they may hinder the immune response against co-infecting agents, thus favoring disease complications (106, 107). In this way, immunomodulation may be a better alternative to immunosuppression, and it may be achieved by neutralizing key cytokines and signaling pathways, rather than inducing a general state of immune hypo-responsiveness.
As IL-6 is a central player in the cytokine release syndrome, blocking its signaling may be a key factor to prevent mortality, lung-tissue damage and ARDS (16). In such an understanding, the a-IL-6R monoclonal antibody Tocilizumab may be an important player on the regulation of such pathological traits (108). Toniati et al. (109), in fact, report an important enhancement on the respiratory functions of 77% of the critically-ill patients (n=100) that received Tocilizumab, as well as radiological improvement in 61% of them. Another medium powered study (n=154) detected a 45% reduction in hazard of death (110), while a smaller study (n=21) detected a decrease in fever, radiological findings (in 90.5% of the patients), CRP (in 84.2%) and oxygen intake (in 75%), along with an improvement in peripheral blood lymphocytes (52.6%) (111).
Moreover, the aIL-1-receptor antagonistic antibody Anakinra has been thought as an alternative to provide downmodulation of the cytokine release, oxidative stress and ACE2 expression. Nonetheless, information about this topic is sparse and comes from rather low-powered studies. For instance, a series of nine cases of patients receiving up to 12 doses of this drug (100mg) showed a modest decrease of CRP and a suspended progression of computed tomography-detected lesions (112), and these observations were further supported by another research team studying 45 patients in 72% of their study subjects (113). Another case series including 11 patients showed that this drug may have certain potential to prevent mechanical ventilation in severe COVID-19 patients when administered early after acute hypoxic respiratory failure onset (114). A bigger study including 96 patients found that this drug reduced both need for invasive mechanical ventilation and mortality among patients with severe forms of COVID-19 (115), and finally one more study (n=93) showed that Anakinra and Tocilizumab are similarly effective at reducing mortality and supplementary oxygenation needs (116).
On the other hand, as the over-expression of PD-1 has been strongly linked to lymphopenia and ultimately to poor outcomes (47), we think that immunostimulation through the blockade of such pathway may be important for controlling viral spread, and thus to enhance patient’s survival. To our notice there are no clinical trials being conducted on the use of Pembrolizumab, Nivolumab or other monoclonal antibodies specific for PD-1 or any of its ligands, but single case reports have shown reductions on CRP, LDH and enhancement of the general state on cancer patients that receive either drug during COVID-19 convalescence (117, 118). Nonetheless, PD-1 blockade has been linked with an enhanced immune response against B and C hepatitis (119, 120).
3.3 The anti-thormbotic axis
In a recent study that included 449 patients with SARS-CoV-2 infections in serious condition who met the criteria for coagulopathy caused by sepsis and with elevated D-dimer levels (higher than 1 μg / ml), it was mentioned that the use of low molecular weight heparin was associated with lower mortality (121); thus the use of anticoagulants improves the prognosis in patients with coagulopathy and high risk of venous thromboembolism. For this reason the International Society of Thrombosis and Haematosis and the American Society of Hematology suggest the prophylactic use of thrombolytic therapy (122). Moreover, low-molecular weight heparin seem to be able to control coagulopathy in the setting of COVID-19, as it has been associated with improvements in lymphocyte counts, D-dimer and CRP levels (123), as well as shorter hospitalization length and lower mortalities (124).
- The antimicrobial axis
As a meta-analysis performed by Lansbury et al. (56) found that only 7% of the hospitalized patients do course with coinfections, and there is no strong data showing that coinfections enhance mortality among COVID-19 patients (125), or that early antibiotic use impacts mortality (126), antibiotics are only recommended when coinfections by bacteria are confirmed. However, empirical antibiotic management is suggested in mechanically ventilated patients with COVID-19 and respiratory failure. Such suggestion comes from extrapolated studies in other viral pneumonias, like influenza (127). On the other hand, in response to lung viral infection, an inflammatory exudate is produced in the alveoli of the lungs, which is the main cause of dyspnea (128), and as such exudate covers the alveoli in a chronic manner the risk of developing bacterial coinfections is enhanced (129). In any case, macrolides, quinolones or β-lactams have been used to treat bacterial coinfections in SARS-CoV-infected patients (130), but these should only be prescribed according to data on the sensitivities of each individual strain.
3.5 The supplementary oxygenation axis
In order to treat the tissue hypoxemia caused by respiratory insufficiency, the supplementary oxygenation therapies seem to be an answer (131). Oxygen administration can be performed in different ways, via face mask or nasal cannula, with high-flow-nasal oxygen or noninvasive ventilation being the most common first-line interventions to improve oxygenation and dyspnea (132).
According to recent studies, therapy with ionized oxygen might be a useful tool to treat COVID-19 patients as in vitro studies confirmed that long-term inhalation of partially positively ionized oxygen (O2+) was associated with less oxidative stress, better ability to secure airway patency and milder pulmonary inflammation response than molecular oxygen (133).
Moreover, the use of common supplementary oxygen has continued, like it is shown by Burn et al. who offered non-invasive pressure support (NIPS) to patients who considered to be too frail to have the potential to benefit from invasive ventilation. They concluded that NIPS had favorable results considering that their population had a poor physiological reserve and had at least the equivalent survival rate in intubated patients from their own institution (134). Another variant used in supplementary oxygen therapy is the position of the patient, Despres et al. suggested that prone positioning combined with high-flow-nasal oxygen or conventional oxygen therapy could be proposed in severe COVID-19 patients, in order to avoid intubation (135). Moreover, a 50% reduction of mortality in patients with severe ARDS was observed when patients were prone positioned in comparison to those in suspine position (136).
- Drug-drug interactions in COVID-19-treatment.
As the treatment of COVID-19 not only requires the use of an effective anti-SARS-CoV-2 agent, but also needs anti-coagulation drugs, anti-inflammatory agents, respiratory support and, in patients receiving respiratory support or with confirmed bacterial coinfections, antibiotics, the potential for presenting drug-drug interactions (DDI) while treating COVID-19 is high. Pharmacodynamic drug-drug interactions occur when the pharmacological effect of one drug is altered by that of another drug in a combination regimen, potentially diminishing or cancelling the effect of one party, or synergizing to cause enhanced damage to the host (137). This problem constitutes the most common medication error as it has a prevalence of 20-40% (138). Despite this, the history of DDI is short as these phenomena were only recognized until 1970, and in 1997 the FDA produced the first documentation for guidance on the conduct of drug prescription and drug metabolism (139).
Currently, many software programs and webpages have been developed to provide information about possible interactions that might exist between drugs, and a very pertinent example is the COVID-19 Drug Interactions (140) platform, which calculates potential DDIs among those that are being assayed in the treatment of such disease.
To our notice, within the most common and severe adverse effects that can occur with anti-COVID-19 medications are:
4.1 Remdesivir
Sufficient data on the compatibility of remdesivir with other intravenous drugs (141) are not available, however the excipient that is formulated to improve its solubility, sulfobutylether-cyclodextrin (SBECD), accumulates in patients with moderate to severe renal dysfunction. Caution is recommended when the drug is administered in combination with other parenteral drugs formulated in SBECD, for example voriconazole and amiodarone, with special emphasis on care in those patients with creatinine clearance of 30 ml/minute. On the other hand, the concomitant use of rifampicin, carbamazepine or phenytoin should be avoided due to decreased exposure to redemsivir (142). Moreover, antagonistic effects between remdesivir and antimalarial drugs (Chloroquine and Hydroxychloroquine)are associated with clathrin-dependent endocytosis, enhanced viral entry/release into host cell, enhanced inflammatory response, pH reduction, negative regulation of kinase activity and enhanced protein tyrosine kinase activity (143).
4.2 Lopinavir/Ritonavir
Lopinavir/Ritonavir interactions occur through various mechanisms, among of which the most important are those involving CYP enzymes, the administration of the drug is contraindicated with flecainide, propafenone, astemizole, terfenadine, ergot derivatives, cisapride, pimozide, midazolam and triazolam, as the metabolism of these drugs is dependent on CYP3A or CYP2D6 (65).
4.3 Umifenovir
Umifenovir is also metabolized by CYP3A4, with a lower contribution of the enzymes P450 and FMO, so that the drug has important interaction with inhibitors and inducers of CYP3A4 (144).
4.4 Favipiravir
Favipiravir is considered an inhibitor of CYP1A2/2C8/2C9/2C19/2D6/2E1/3A4, OAT1 and OAT3 (145), and has a liver metabolism through AO (146). A substrate of CYP2C8 is zoplicone, so caution should be exercised when administered with favipiravir (145) some pharmaco-inhibitors of AO are raloxifene, tamoxifen, estradiol, cimetidine, felodipine, amlodipine, verapamil, propafenone and amitriptyline, however no relevant clinical interaction based on AO inhibition is established yet (146). Moreover, the administration of favipirivir increases acetaminophen accumulation (146) and inhibits the formation of acetaminophen sulfate (147), so that combining both drugs is not recommended (146).
4.5 Chloroquine/ Hydroxichloroquine
As these drugs are metabolized through CYP2D6, the inhibitors or inducers of such enzyme should be avoided. Moreover, its combination with Lopinavir/Ritonavir may increase QTc intervals, or cause hypoglycemia (143). Extreme care must be undertaken in its administration in patients with pre-existing heart disease and in its use for the treatment of COVID-19 due to the affectation caused by heart disease. Moreover, as it is metabolized through cytochrome P450, its use in conjunction with azithromycin, which inhibits CYP450, causes a reduction in the metabolism of hydroxychloroquine causing toxic accumulation of the drug (148), and this combination also prolongs QTc intervals.
Fluoxetine, paroxetine, amitriptyline, bupropion and duloxetine may increase both drugs’ accumulation, as they are inhibitors of the enzymes CYP3A4 and CYP2D6, while a CYP3A4 inducer such as carbamazepine, may cause reduced levels of Chloroquine and Hydroxychloroquine.
In such panorama, especial care should be taken to avoid these interactions, which may further complicate the pathology.
- Conclusions
The emergency that the COVID-19 pandemic represents has impacted in the field of medical and biomedical research, imposing an urgent need for safe, effective and widely-available drugs. In this way, drug repurposing has been a staple in anti-COVID-19 therapy, where similarities in the form and function of COVID-19 with other viruses and viral diseases have been key to find plausible therapeutic pathways. In this way, we think that such disease’s pathophysiology is comprised of five main axes: 1) the respiratory disfunction, 2) the inflammatory dysregulation, 3) coagulopathy, 4) coinfections and 5) last but not least, the viral replication.
In concordance to this understanding, studies on 1) type of respiratory support and positioning of the patient, 2) immunosuppressant and immunomodulatory drugs, 3) anticoagulants, 4) antimicotics and antibiotics and 5) anti-viral agents have been performed to conform a rapidly increasing body of evidence to lead the COVID-19 therapeutics. Nonetheless, vaster and stronger data is needed regarding the therapeutics for each one of these axes, especially in regards to data collected from critically ill patients, patients with relevant comorbidities and in the prophylactic use of antiviral drugs.
If data about the efficacy of the aforementioned drugs in the treatment of COVID-19 is sparse, it is even sparser the information regarding their safety when used in combination. In such an understanding, we think that the whole role of adjunctive therapy requires more research in terms of its efficacy and safety.
- Acknowledgements
All the authors wish to thank Sociedad Española de Beneficencia (Pachuca, Hgo), for providing the technical and financial means for the production and publication of this article.
- Conflicts of interest
None of the authors have any conflicts of interest to declare.
- Data availability statement
As the present manuscript is a review article there is no relevant information to be made publicly available. Whenever possible, readers should refer to the proper references.
- References
1. Organization WH. 2020. WHO Coronavirus Disease (COVID-19) Dashboard. World Health Organization
2. (OCDE) OfEC-oaD. 2020. OECD Economic Outlook, .
3. Remes-Troche JM, Ramos-de-la-Medina A, Manríquez-Reyes M, Martínez-Pérez-Maldonado L, Lara EL, Solís-González MA. 2020. Initial Gastrointestinal Manifestations in Patients with SARS-CoV-2 in 112 patients from Veracruz (Southeastern Mexico). Gastroenterology
4. Guo YR, Cao QD, Hong ZS, Tan YY, Chen SD, Jin HJ, Tan KS, Wang DY, Yan Y. 2020. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil Med Res 7: 11
5. Pascarella G, Strumia A, Piliego C, Bruno F, Del Buono R, Costa F, Scarlata S, Agro FE. 2020. COVID-19 diagnosis and management: a comprehensive review. J Intern Med
6. Salvi R, Patankar P. 2020. Emerging pharmacotherapies for COVID-19. Biomed Pharmacother 128: 110267
7. Liu F, Xu A, Zhang Y, Xuan W, Yan T, Pan K, Yu W, Zhang J. 2020. Patients of COVID-19 may benefit from sustained Lopinavir-combined regimen and the increase of Eosinophil may predict the outcome of COVID-19 progression. Int J Infect Dis 95: 183-91
8. Zhu Z, Lu Z, Xu T, Chen C, Yang G, Zha T, Lu J, Xue Y. 2020. Arbidol monotherapy is superior to lopinavir/ritonavir in treating COVID-19. J Infect 81: e21-e3
9. Cai Q, Yang M, Liu D, Chen J, Shu D, Xia J, Liao X, Gu Y, Cai Q, Yang Y, Shen C, Li X, Peng L, Huang D, Zhang J, Zhang S, Wang F, Liu J, Chen L, Chen S, Wang Z, Zhang Z, Cao R, Zhong W, Liu Y, Liu L. 2020. Experimental Treatment with Favipiravir for COVID-19: An Open-Label Control Study. Engineering (Beijing)
10. Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J, Giordanengo V, Vieira VE, Dupont HT, Honoré S, Colson P, Chabrière E, La Scola B, Rolain JM, Brouqui P, Raoult D. 2020. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents: 105949
11. Feeney E, Wallace D, Cotter A, Tinago W, McCarthy C, Keane D, Hussain R, Alvarez Barco E, Doran P, Mallon P. 2020. The COVIRL-001 Trial: A multicentre, prospective, randomised trial comparing standard of care (SOC) alone, SOC plus hydroxychloroquine monotherapy or SOC plus a combination of hydroxychloroquine and azithromycin in the treatment of non- critical, SARS-CoV-2 PCR-positive population not requiring immediate resuscitation or ventilation but who have evidence of clinical decline: A structured summary of a study protocol for a randomised controlled trial. Trials 21: 430
12. Caly L, Druce JD, Catton MG, Jans DA, Wagstaff KM. 2020. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res 178: 104787
13. Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, Fu S, Gao L, Cheng Z, Lu Q, Hu Y, Luo G, Wang K, Lu Y, Li H, Wang S, Ruan S, Yang C, Mei C, Wang Y, Ding D, Wu F, Tang X, Ye X, Ye Y, Liu B, Yang J, Yin W, Wang A, Fan G, Zhou F, Liu Z, Gu X, Xu J, Shang L, Zhang Y, Cao L, Guo T, Wan Y, Qin H, Jiang Y, Jaki T, Hayden FG, Horby PW, Cao B, Wang C. 2020. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 395: 1569-78
14. Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC. 2020. Remdesivir for the Treatment of Covid-19 - Preliminary Report.
15. Luo P, Liu Y, Qiu L, Liu X, Liu D, Li J. 2020. Tocilizumab treatment in COVID-19: A single center experience. 92: 814-8
16. Zhang C, Wu Z, Li JW, Zhao H, Wang GQ. 2020. Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int J Antimicrob Agents 55: 105954
17. Viale P, Bartoletti M. 2020. Clinical experience with therapeutic dose of Low-Molecular-Weight Heparin. Infez Med 28: 118-21
18. therapy REoC-. 2020. Wellcome - RECOVERY Trial.
19. Zhou M, Zhang X, Qu J. 2020. Coronavirus disease 2019 (COVID-19): a clinical update. Front Med 14: 126-35
20. Zhang Y, Xiao M, Zhang S, Xia P, Cao W, Jiang W, Chen H, Ding X, Zhao H, Zhang H, Wang C, Zhao J, Sun X, Tian R, Wu W, Wu D, Ma J, Chen Y, Zhang D, Xie J, Yan X, Zhou X, Liu Z, Wang J, Du B, Qin Y, Gao P, Qin X, Xu Y, Zhang W, Li T, Zhang F, Zhao Y, Li Y, Zhang S. 2020. Coagulopathy and Antiphospholipid Antibodies in Patients with Covid-19. N Engl J Med 382: e38
21. Iba T, Levy JH, Levi M, Thachil J. 2020. Coagulopathy in COVID-19. J Thromb Haemost
22. Baltimore D. 1971. Expression of animal virus genomes. Bacteriol Rev 35: 235-41
23. Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. 2020. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 181: 271-80.e8
24. Li MY, Li L, Zhang Y, Wang XS. 2020. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty 9: 45
25. Yuki K, Fujiogi M, Koutsogiannaki S. 2020. COVID-19 pathophysiology: A review. Clin Immunol 215: 108427
26. Lukassen S, Chua RL, Trefzer T, Kahn NC, Schneider MA, Muley T, Winter H, Meister M, Veith C, Boots AW, Hennig BP, Kreuter M. 2020. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells. 39: e105114
27. Ziegler CGK, Allon SJ, Nyquist SK, Mbano IM, Miao VN, Tzouanas CN, Cao Y, Yousif AS, Bals J, Hauser BM, Feldman J, Muus C, Wadsworth MH, 2nd, Kazer SW, Hughes TK, Doran B, Gatter GJ, Vukovic M, Taliaferro F, Mead BE, Guo Z, Wang JP, Gras D, Plaisant M, Ansari M, Angelidis I, Adler H, Sucre JMS, Taylor CJ, Lin B, Waghray A, Mitsialis V, Dwyer DF, Buchheit KM, Boyce JA, Barrett NA, Laidlaw TM, Carroll SL, Colonna L, Tkachev V, Peterson CW, Yu A, Zheng HB, Gideon HP, Winchell CG, Lin PL, Bingle CD, Snapper SB, Kropski JA, Theis FJ, Schiller HB, Zaragosi LE, Barbry P, Leslie A, Kiem HP, Flynn JL, Fortune SM, Berger B, Finberg RW, Kean LS, Garber M, Schmidt AG, Lingwood D, Shalek AK, Ordovas-Montanes J. 2020. SARS-CoV-2 Receptor ACE2 Is an Interferon-Stimulated Gene in Human Airway Epithelial Cells and Is Detected in Specific Cell Subsets across Tissues. Cell 181: 1016-35.e19
28. Sajuthi SP, DeFord P, Jackson ND, Montgomery MT, Everman JL, Rios CL, Pruesse E, Nolin JD, Plender EG, Wechsler ME, Mak AC, Eng C, Salazar S, Medina V, Wohlford EM, Huntsman S, Nickerson DA, Germer S, Zody MC, Abecasis G, Kang HM, Rice KM, Kumar R, Oh S, Rodriguez-Santana J, Burchard EG, Seibold MA. 2020. Type 2 and interferon inflammation strongly regulate SARS-CoV-2 related gene expression in the airway epithelium. bioRxiv
29. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. 2004. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203: 631-7
30. Jia HP, Look DC, Shi L, Hickey M, Pewe L, Netland J, Farzan M, Wohlford-Lenane C, Perlman S, McCray PB, Jr. 2005. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J Virol 79: 14614-21
31. Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. 2020. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med 8: 420-2
32. Zhang H, Zhou P, Wei Y, Yue H, Wang Y, Hu M, Zhang S, Cao T, Yang C, Li M, Guo G, Chen X, Chen Y, Lei M, Liu H, Zhao J, Peng P, Wang CY, Du R. 2020. Histopathologic Changes and SARS-CoV-2 Immunostaining in the Lung of a Patient With COVID-19. Ann Intern Med 172: 629-32
33. Yao XH, He ZC, Li TY, Zhang HR, Wang Y, Mou H, Guo Q, Yu SC, Ding Y, Liu X, Ping YF, Bian XW. 2020. Pathological evidence for residual SARS-CoV-2 in pulmonary tissues of a ready-for-discharge patient. Cell Res 30: 541-3
34. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS, China Medical Treatment Expert Group for C. 2020. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med 382: 1708-20
35. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. 2020. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395: 507-13
36. Li X, Ma X. 2020. Acute respiratory failure in COVID-19: is it "typical" ARDS? 24: 198
37. Li X, Ma X. 2020. Acute respiratory failure in COVID-19: is it "typical" ARDS? Crit Care 24: 198
38. Zhang X, Li S, Niu S. 2020. ACE2 and COVID-19 and the resulting ARDS. Postgrad Med J 96: 403-7
39. Vardhana SA, Wolchok JD. 2020. The many faces of the anti-COVID immune response. J Exp Med 217
40. Wen W, Su W, Tang H, Le W, Zhang X, Zheng Y, Liu X, Xie L, Li J, Ye J, Cui X, Miao Y, Wang D, Dong J, Xiao C-L, Chen W, Wang H. 2020. Immune Cell Profiling of COVID-19 Patients in the Recovery Stage by Single-Cell Sequencing. medRxiv: 2020.03.23.20039362
41. Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, Wang T, Zhang X, Chen H, Yu H, Zhang X, Zhang M, Wu S, Song J, Chen T, Han M, Li S, Luo X, Zhao J, Ning Q. 2020. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest 130: 2620-9
42. Hu B, Huang S, Yin L. 2020. The cytokine storm and COVID-19. J Med Virol
43. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497-506
44. Chen L, Liu HG, Liu W, Liu J, Liu K, Shang J, Deng Y, Wei S. 2020. [Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia]. Zhonghua Jie He He Hu Xi Za Zhi 43: E005
45. Bordoni V, Sacchi A, Cimini E, Notari S, Grassi G, Tartaglia E, Casetti R, Giancola L, Bevilacqua N, Maeurer M, Zumla A, Locatelli F, De Benedetti F, Palmieri F, Marchioni L, Capobianchi MR, D'Offizi G, Petrosillo N, Antinori A, Nicastri E, Ippolito G, Agrati C. 2020. An inflammatory profile correlates with decreased frequency of cytotoxic cells in COVID-19. Clin Infect Dis
46. Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Moller R, Jordan TX, Oishi K, Panis M, Sachs D, Wang TT, Schwartz RE, Lim JK, Albrecht RA, tenOever BR. 2020. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell 181: 1036-45 e9
47. Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, Chen L, Li M, Liu Y, Wang G, Yuan Z, Feng Z, Zhang Y, Wu Y, Chen Y. 2020. Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19). Frontiers in Immunology 11
48. Guo L, Ren L, Yang S, Xiao M, Chang, Yang F, Dela Cruz CS, Wang Y, Wu C, Xiao Y, Zhang L, Han L, Dang S, Xu Y, Yang Q, Xu S, Zhu H, Xu Y, Jin Q, Sharma L, Wang L, Wang J. 2020. Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin Infect Dis
49. Tang F, Quan Y, Xin ZT, Wrammert J, Ma MJ, Lv H, Wang TB, Yang H, Richardus JH, Liu W, Cao WC. 2011. Lack of peripheral memory B cell responses in recovered patients with severe acute respiratory syndrome: a six-year follow-up study. J Immunol 186: 7264-8
50. Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS. 2020. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis
51. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. 2020. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054-62
52. Whyte CS, Morrow GB, Mitchell JL, Chowdary P, Mutch NJ. 2020. Fibrinolytic abnormalities in acute respiratory distress syndrome (ARDS) and versatility of thrombolytic drugs to treat COVID-19. J Thromb Haemost 18: 1548-55
53. Connors JM, Levy JH. 2020. COVID-19 and its implications for thrombosis and anticoagulation. Blood 135: 2033-40
54. Iba T, Levy JH, Connors JM, Warkentin TE, Thachil J, Levi M. 2020. The unique characteristics of COVID-19 coagulopathy. Crit Care 24: 360
55. Zhu X, Ge Y, Wu T, Zhao K, Chen Y, Wu B, Zhu F, Zhu B, Cui L. 2020. Co-infection with respiratory pathogens among COVID-2019 cases. Virus Res 285: 198005
56. Lansbury L, Lim B, Baskaran V, Lim WS. 2020. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect 81: 266-75
57. Lai CC, Wang CY, Hsueh PR. 2020. Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J Microbiol Immunol Infect 53: 505-12
58. Garcia LF. 2020. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front Immunol 11: 1441
59. Lipsky MS, Sharp LK. 2001. From idea to market: the drug approval process. J Am Board Fam Pract 14: 362-7
60. Kumar R, Harilal S, Gupta SV, Jose J, Thomas Parambi DG, Uddin MS, Shah MA, Mathew B. 2019. Exploring the new horizons of drug repurposing: A vital tool for turning hard work into smart work. Eur J Med Chem 182: 111602
61. Wu R, Wang L, Kuo HD, Shannar A, Peter R, Chou PJ, Li S, Hudlikar R, Liu X, Liu Z, Poiani GJ, Amorosa L, Brunetti L, Kong AN. 2020. An Update on Current Therapeutic Drugs Treating COVID-19. Curr Pharmacol Rep: 1-15
62. Lu CC, Chen MY, Lee WS, Chang YL. 2020. Potential therapeutic agents against COVID-19: What we know so far. J Chin Med Assoc 83: 534-6
63. Li Z, Wang X, Cao D, Sun R, Li C, Li G. 2020. Rapid review for the anti-coronavirus effect of remdesivir. Drug Discov Ther 14: 73-6
64. Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, Feldt T, Green G, Green ML, Lescure FX, Nicastri E. 2020. Compassionate Use of Remdesivir for Patients with Severe Covid-19. 382: 2327-36
65. Cvetkovic RS, Goa KL. 2003. Lopinavir/ritonavir: a review of its use in the management of HIV infection. Drugs 63: 769-802
66. Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. 2020. Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review. Jama
67. Ul Qamar MT, Alqahtani SM, Alamri MA, Chen LL. 2020. Structural basis of SARS-CoV-2 3CL(pro) and anti-COVID-19 drug discovery from medicinal plants. J Pharm Anal
68. Nutho B, Mahalapbutr P, Hengphasatporn K, Pattaranggoon NC, Simanon N, Shigeta Y, Hannongbua S, Rungrotmongkol T. 2020. Why Are Lopinavir and Ritonavir Effective against the Newly Emerged Coronavirus 2019? Atomistic Insights into the Inhibitory Mechanisms. Biochemistry 59: 1769-79
69. Ye XT, Luo YL, Xia SC, Sun QF, Ding JG, Zhou Y, Chen W, Wang XF, Zhang WW, Du WJ, Ruan ZW, Hong L. 2020. Clinical efficacy of lopinavir/ritonavir in the treatment of Coronavirus disease 2019. Eur Rev Med Pharmacol Sci 24: 3390-6
70. Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, Ruan L, Song B, Cai Y, Wei M, Li X, Xia J, Chen N, Xiang J, Yu T, Bai T, Xie X, Zhang L, Li C, Yuan Y, Chen H, Li H, Huang H, Tu S, Gong F, Liu Y, Wei Y, Dong C, Zhou F, Gu X, Xu J, Liu Z, Zhang Y, Li H, Shang L, Wang K, Li K, Zhou X, Dong X, Qu Z, Lu S, Hu X, Ruan S, Luo S, Wu J, Peng L, Cheng F, Pan L, Zou J, Jia C, Wang J, Liu X, Wang S, Wu X, Ge Q, He J, Zhan H, Qiu F, Guo L, Huang C, Jaki T, Hayden FG, Horby PW, Zhang D, Wang C. 2020. A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19. N Engl J Med 382: 1787-99
71. Hung IF, Lung KC, Tso EY, Liu R, Chung TW, Chu MY, Ng YY, Lo J, Chan J, Tam AR, Shum HP, Chan V, Wu AK, Sin KM, Leung WS, Law WL, Lung DC, Sin S, Yeung P, Yip CC, Zhang RR, Fung AY, Yan EY, Leung KH, Ip JD, Chu AW, Chan WM, Ng AC, Lee R, Fung K, Yeung A, Wu TC, Chan JW, Yan WW, Chan WM, Chan JF, Lie AK, Tsang OT, Cheng VC, Que TL, Lau CS, Chan KH, To KK, Yuen KY. 2020. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet 395: 1695-704
72. Huang Y-Q, Tang S-Q, Xu X-L, Zeng Y-M, He X-Q, Li Y, Harypursat V, Lu Y-Q, Wan Y, Zhang L, Sun Q-Z, Sun N-N, Wang G-X, Yang Z-P, Chen Y-K. 2020. No Statistically Apparent Difference in Antiviral Effectiveness Observed Among Ribavirin Plus Interferon-Alpha, Lopinavir/Ritonavir Plus Interferon-Alpha, and Ribavirin Plus Lopinavir/Ritonavir Plus Interferon-Alpha in Patients With Mild to Moderate Coronavirus Disease 2019: Results of a Randomized, Open-Labeled Prospective Study. Frontiers in Pharmacology 11
73. Boriskin YS, Leneva IA, Pécheur EI, Polyak SJ. 2008. Arbidol: a broad-spectrum antiviral compound that blocks viral fusion. Curr Med Chem 15: 997-1005
74. Pecheur EI, Borisevich V, Halfmann P, Morrey JD, Smee DF, Prichard M, Mire CE, Kawaoka Y, Geisbert TW, Polyak SJ. 2016. The Synthetic Antiviral Drug Arbidol Inhibits Globally Prevalent Pathogenic Viruses. J Virol 90: 3086-92
75. Villalain J. 2010. Membranotropic effects of arbidol, a broad anti-viral molecule, on phospholipid model membranes. J Phys Chem B 114: 8544-54
76. Vankadari N. 2020. Arbidol: A potential antiviral drug for the treatment of SARS-CoV-2 by blocking trimerization of the spike glycoprotein. Int J Antimicrob Agents: 105998
77. Lian N, Xie H, Lin S, Huang J, Zhao J, Lin Q. 2020. Umifenovir treatment is not associated with improved outcomes in patients with coronavirus disease 2019: a retrospective study. Clin Microbiol Infect 26: 917-21
78. Siordia Jr JA, Bernaba M, Yoshino K, Ulhaque A, Kumar S, Bernaba M, Bergin E. 2020. Systematic and Statistical Review of Coronavirus Disease 19 Treatment Trials. SN Compr Clin Med 2020: 1-12
79. Yang C, Ke C, Yue D, Li W, Hu Z, Liu W, Hu S, Wang S, Liu J. 2020. Effectiveness of Arbidol for COVID-19 Prevention in Health Professionals. Front Public Health 8: 249
80. Furuta Y, Gowen BB, Takahashi K, Shiraki K, Smee DF, Barnard DL. 2013. Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antiviral Res 100: 446-54
81. Bai CQ, Mu JS, Kargbo D, Song YB, Niu WK, Nie WM, Kanu A, Liu WW, Wang YP, Dafae F, Yan T, Hu Y, Deng YQ, Lu HJ, Yang F, Zhang XG, Sun Y, Cao YX, Su HX, Sun Y, Liu WS, Wang CY, Qian J, Liu L, Wang H, Tong YG, Liu ZY, Chen YS, Wang HQ, Kargbo B, Gao GF, Jiang JF. 2016. Clinical and Virological Characteristics of Ebola Virus Disease Patients Treated With Favipiravir (T-705)-Sierra Leone, 2014. Clin Infect Dis 63: 1288-94
82. Shiraki K, Daikoku T. 2020. Favipiravir, an anti-influenza drug against life-threatening RNA virus infections. Pharmacol Ther 209: 107512
83. Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, Shi Z, Hu Z. 2020. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. 30: 269-71
84. Wagstaff KM, Sivakumaran H, Heaton SM, Harrich D, Jans DA. 2012. Ivermectin is a specific inhibitor of importin α/β-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus. Biochem J 443: 851-6
85. Barrows NJ, Campos RK, Powell ST, Prasanth KR, Schott-Lerner G, Soto-Acosta R, Galarza-Muñoz G, McGrath EL, Urrabaz-Garza R, Gao J, Wu P, Menon R, Saade G, Fernandez-Salas I, Rossi SL, Vasilakis N, Routh A, Bradrick SS, Garcia-Blanco MA. 2016. A Screen of FDA-Approved Drugs for Inhibitors of Zika Virus Infection. Cell Host Microbe 20: 259-70
86. Varghese FS, Kaukinen P, Gläsker S, Bespalov M, Hanski L, Wennerberg K, Kümmerer BM, Ahola T. 2016. Discovery of berberine, abamectin and ivermectin as antivirals against chikungunya and other alphaviruses. Antiviral Res 126: 117-24
87. Chaccour C, Hammann F, Ramón-García S, Rabinovich NR. 2020. Ivermectin and COVID-19: Keeping Rigor in Times of Urgency. Am J Trop Med Hyg 102: 1156-7
88. Organization PAH. 2020. Ongoing Living Update of Potential COVID-19 Therapeutics: summary of rapid systematic reviews. Pan American Health Organization
89. Organization WH. 2020. “Solidarity” clinical trial for COVID-19 treatments
90. Schultz KR, Gilman AL. 1997. The lysosomotropic amines, chloroquine and hydroxychloroquine: a potentially novel therapy for graft-versus-host disease. Leuk Lymphoma 24: 201-10
91. Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, Shi Z, Hu Z, Zhong W, Xiao G. 2020. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 30: 269-71
92. Savarino A, Di Trani L, Donatelli I, Cauda R, Cassone A. 2006. New insights into the antiviral effects of chloroquine. Lancet Infect Dis 6: 67-9
93. Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, Li Y, Hu Z. 2020. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. 6: 16
94. Devaux CA, Rolain JM, Colson P, Raoult D. 2020. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int J Antimicrob Agents 55: 105938
95. Silva JC, Mariz HA, Rocha LF, Jr., Oliveira PS, Dantas AT, Duarte AL, Pitta Ida R, Galdino SL, Pitta MG. 2013. Hydroxychloroquine decreases Th17-related cytokines in systemic lupus erythematosus and rheumatoid arthritis patients. Clinics (Sao Paulo) 68: 766-71
96. Cao X. 2020. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol 20: 269-70
97. Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J, Giordanengo V, Vieira VE, Tissot Dupont H, Honoré S, Colson P, Chabrière E, La Scola B, Rolain JM, Brouqui P, Raoult D. 2020. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents: 105949
98. Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Sevestre J, Mailhe M, Doudier B, Aubry C, Amrane S, Seng P, Hocquart M, Eldin C, Finance J, Vieira VE, Tissot-Dupont HT, Honoré S, Stein A, Million M, Colson P, La Scola B, Veit V, Jacquier A, Deharo JC, Drancourt M, Fournier PE, Rolain JM, Brouqui P, Raoult D. 2020. Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: A pilot observational study. Travel Med Infect Dis 34: 101663
99. Million M, Lagier JC, Gautret P, Colson P, Fournier PE, Amrane S, Hocquart M, Mailhe M, Esteves-Vieira V, Doudier B, Aubry C, Correard F, Giraud-Gatineau A, Roussel Y, Berenger C, Cassir N, Seng P, Zandotti C, Dhiver C, Ravaux I, Tomei C, Eldin C, Tissot-Dupont H, Honoré S, Stein A, Jacquier A, Deharo JC, Chabrière E, Levasseur A, Fenollar F, Rolain JM, Obadia Y, Brouqui P, Drancourt M, La Scola B, Parola P, Raoult D. 2020. Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: A retrospective analysis of 1061 cases in Marseille, France. Travel Med Infect Dis 35: 101738
100. Geleris J, Sun Y, Platt J, Zucker J, Baldwin M, Hripcsak G, Labella A, Manson DK, Kubin C, Barr RG, Sobieszczyk ME, Schluger NW. 2020. Observational Study of Hydroxychloroquine in Hospitalized Patients with Covid-19. New England Journal of Medicine 382: 2411-8
101. Mahevas M, Tran VT, Roumier M, Chabrol A, Paule R, Guillaud C, Fois E, Lepeule R, Szwebel TA, Lescure FX, Schlemmer F, Matignon M, Khellaf M, Crickx E, Terrier B, Morbieu C, Legendre P, Dang J, Schoindre Y, Pawlotsky JM, Michel M, Perrodeau E, Carlier N, Roche N, de Lastours V, Ourghanlian C, Kerneis S, Menager P, Mouthon L, Audureau E, Ravaud P, Godeau B, Gallien S, Costedoat-Chalumeau N. 2020. Clinical efficacy of hydroxychloroquine in patients with covid-19 pneumonia who require oxygen: observational comparative study using routine care data. BMJ 369: m1844
102. Sevilla-Castillo F, Roque-Reyes OJ, Romero-Lechuga F, Gomez-Nunez MF, Castillo-Lopez M, Medina-Santos D, Roman PO, Flores-Hernandez JR, Mendez-Coca JD, Montano-Olmos D, Farfan-Lazos KC, Tobon-Cubillos M, Viveros-Hernandez A, Torres-Ortega L, Hernandez-Skewes KY, Montiel-Bravo G, Ortega-Rodriguez S, Peon AN. 2021. Both Chloroquine and Lopinavir/Ritonavir Are Ineffective for COVID-19 Treatment and Combined Worsen the Pathology: A Single-Center Experience with Severely Ill Patients. Biomed Res Int 2021: 8821318
103. Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L. 2020. Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report.
104. Sterne JAC, Murthy S, Diaz JV, Slutsky AS, Villar J, Angus DC, Annane D, Azevedo LCP, Berwanger O, Cavalcanti AB, Dequin PF, Du B, Emberson J, Fisher D, Giraudeau B, Gordon AC, Granholm A, Green C, Haynes R, Heming N, Higgins JPT, Horby P, Jüni P, Landray MJ, Le Gouge A, Leclerc M, Lim WS, Machado FR, McArthur C, Meziani F, Møller MH, Perner A, Petersen MW, Savovic J, Tomazini B, Veiga VC, Webb S, Marshall JC. 2020. Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis. Jama
105. Li Q, Li W, Jin Y, Xu W, Huang C, Li L, Huang Y, Fu Q, Chen L. 2020. Efficacy Evaluation of Early, Low-Dose, Short-Term Corticosteroids in Adults Hospitalized with Non-Severe COVID-19 Pneumonia: A Retrospective Cohort Study. Infect Dis Ther
106. Johnson RM, Vinetz JM. 2020. Dexamethasone in the management of covid -19. Bmj 370: m2648
107. Theoharides TC, Conti P. 2020. Dexamethasone for COVID-19? Not so fast. J Biol Regul Homeost Agents 34
108. Fu B, Xu X, Wei H. 2020. Why tocilizumab could be an effective treatment for severe COVID-19? 18: 164
109. Toniati P, Piva S, Cattalini M, Garrafa E, Regola F, Castelli F, Franceschini F, Airò P, Bazzani C, Beindorf EA, Berlendis M, Bezzi M, Bossini N, Castellano M, Cattaneo S, Cavazzana I, Contessi GB, Crippa M, Delbarba A, De Peri E, Faletti A, Filippini M, Filippini M, Frassi M, Gaggiotti M, Gorla R, Lanspa M, Lorenzotti S, Marino R, Maroldi R, Metra M, Matteelli A, Modina D, Moioli G, Montani G, Muiesan ML, Odolini S, Peli E, Pesenti S, Pezzoli MC, Pirola I, Pozzi A, Proto A, Rasulo FA, Renisi G, Ricci C, Rizzoni D, Romanelli G, Rossi M, Salvetti M, Scolari F, Signorini L, Taglietti M, Tomasoni G, Tomasoni LR, Turla F, Valsecchi A, Zani D, Zuccalà F, Zunica F, Focà E, Andreoli L, Latronico N. 2020. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmun Rev 19: 102568
110. Somers EC, Eschenauer GA, Troost JP, Golob JL, Gandhi TN, Wang L, Zhou N, Petty LA, Baang JH, Dillman NO, Frame D, Gregg KS, Kaul DR, Nagel J, Patel TS, Zhou S, Lauring AS, Hanauer DA, Martin E, Sharma P, Fung CM, Pogue JM. 2020. Tocilizumab for treatment of mechanically ventilated patients with COVID-19. medRxiv
111. Xu X, Han M. 2020. Effective treatment of severe COVID-19 patients with tocilizumab. 117: 10970-5
112. Aouba A, Baldolli A, Geffray L, Verdon R, Bergot E, Martin-Silva N, Justet A. 2020. Targeting the inflammatory cascade with anakinra in moderate to severe COVID-19 pneumonia: case series. Ann Rheum Dis
113. Cavalli G, De Luca G, Campochiaro C, Della-Torre E, Ripa M, Canetti D, Oltolini C, Castiglioni B, Tassan Din C, Boffini N, Tomelleri A, Farina N, Ruggeri A, Rovere-Querini P, Di Lucca G, Martinenghi S, Scotti R, Tresoldi M, Ciceri F, Landoni G, Zangrillo A, Scarpellini P, Dagna L. 2020. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. Lancet Rheumatol 2: e325-e31
114. Navarro-Millán I, Sattui SE. 2020. Use of Anakinra to Prevent Mechanical Ventilation in Severe COVID-19: A Case Series.
115. Huet T, Beaussier H, Voisin O, Jouveshomme S, Dauriat G, Lazareth I, Sacco E, Naccache JM, Bézie Y, Laplanche S, Le Berre A, Le Pavec J, Salmeron S, Emmerich J, Mourad JJ, Chatellier G, Hayem G. 2020. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol 2: e393-e400
116. Langer-Gould A, Smith JB, Gonzales EG, Castillo RD, Garza Figueroa J, Ramanathan A, Li BH, Gould MK. 2020. Early Identification of COVID-19 Cytokine Storm and Treatment with Anakinra or Tocilizumab. Int J Infect Dis
117. Yekeduz E, Dursun B, Aydin GC, Yazgan SC, Ozturk HH, Azap A, Utkan G, Urun Y. 2020. Clinical course of COVID-19 infection in elderly patient with melanoma on nivolumab. J Oncol Pharm Pract 26: 1289-94
118. O'Kelly B, McGettrick P, Angelov D, Fay M, McGinty T, Cotter AG, Sheehan G, Lambert JS. 2020. Outcome of a patient with refractory Hodgkin lymphoma on pembrolizumab, infected with SARS-CoV-2. Br J Haematol 190: e1-e3
119. Gane E, Verdon DJ, Brooks AE, Gaggar A, Nguyen AH, Subramanian GM, Schwabe C, Dunbar PR. 2019. Anti-PD-1 blockade with nivolumab with and without therapeutic vaccination for virally suppressed chronic hepatitis B: A pilot study. J Hepatol 71: 900-7
120. Moreno-Cubero E, Larrubia JR. 2016. Specific CD8(+) T cell response immunotherapy for hepatocellular carcinoma and viral hepatitis. World J Gastroenterol 22: 6469-83
121. Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. 2020. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 18: 1094-9
122. Thachil J, Tang N, Gando S. 2020. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. 18: 1023-6
123. Yormaz B, ErgÜn D, TÜlek B, ErgÜn R, Arslan U, Kanat F. 2020. The impact of the "low molecular weight heparin" administration on the clinical course of COVID-19 disease. Turk J Med Sci
124. Paolisso P, Bergamaschi L, D'Angelo EC, Donati F, Giannella M, Tedeschi S, Pascale R, Bartoletti M, Tesini G, Biffi M, Cosmi B, Pizzi C, Viale P, Galié N. 2020. Preliminary Experience With Low Molecular Weight Heparin Strategy in COVID-19 Patients. Front Pharmacol 11: 1124
125. OPS/OMS. 2020. Actualización Epidemiológica: Nuevo coronavirus (COVID-19). ed. OPdlSOMdl Salud. Washington, D.C.: Organización Panamericana de la Salud/Organización Mundial de la Salud
126. Buetti N, Mazzuchelli T, Lo Priore E, Balmelli C, Llamas M, Pallanza M, Elzi L, Consonni V, Trimboli P, Forni-Ogna V, Bernasconi E. 2020. Early administered antibiotics do not impact mortality in critically ill patients with COVID-19. J Infect 81: e148-e9
127. Alhazzani W, Møller MH, Arabi YM, Loeb M, Gong MN, Fan E, Oczkowski S, Levy MM, Derde L, Dzierba A, Du B, Aboodi M, Wunsch H, Cecconi M, Koh Y, Chertow DS, Maitland K, Alshamsi F, Belley-Cote E, Greco M, Laundy M, Morgan JS, Kesecioglu J, McGeer A, Mermel L, Mammen MJ, Alexander PE, Arrington A, Centofanti JE, Citerio G, Baw B, Memish ZA, Hammond N, Hayden FG, Evans L, Rhodes A. 2020. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Medicine 46: 854-87
128. Gianotti R, Veraldi S, Recalcati S, Cusini M, Ghislanzoni M, Boggio F, Fox LP. 2020. Cutaneous Clinico-Pathological Findings in three COVID-19-Positive Patients Observed in the Metropolitan Area of Milan, Italy. Acta Derm Venereol 100: adv00124
129. Perinel S, Launay M, Botelho-Nevers E, Diconne E, Louf-Durier A, Lachand R, Murgier M, Page D, Vermesch R, Thierry G, Delavenne X. 2020. Towards Optimization of Hydroxychloroquine Dosing in Intensive Care Unit COVID-19 Patients. Clin Infect Dis
130. Wang J, Tang Y, Ma Y, Zhou Q, Li W, Baskota M, Yang Y, Wang X, Li Q, Luo X, Fukuoka T, Ahn HS, Lee MS, Luo Z, Liu E, Chen Y. 2020. Efficacy and safety of antibiotic agents in children with COVID-19: a rapid review. Ann Transl Med 8: 619
131. Gu WJ, Zhang Z, Van Poucke S. 2017. Oxygen Therapy and Ventilatory Support. Can Respir J 2017: 2462818
132. Woyke S, Rauch S, Strohle M, Gatterer H. 2020. Modulation of Hb-O2 affinity to improve hypoxemia in COVID-19 patients. Clin Nutr
133. Perecinsky S, Donic V, Legath L. 2020. Partially ionized medical oxygen as a supplementary treatment for COVID-19. Wien Klin Wochenschr
134. Burns GP, Lane ND, Tedd HM, Deutsch E, Douglas F, West SD, Macfarlane JG, Wiscombe S, Funston W. 2020. Improved survival following ward-based non-invasive pressure support for severe hypoxia in a cohort of frail patients with COVID-19: retrospective analysis from a UK teaching hospital. BMJ Open Respir Res 7
135. Despres C, Brunin Y, Berthier F, Pili-Floury S, Besch G. 2020. Prone positioning combined with high-flow nasal or conventional oxygen therapy in severe Covid-19 patients. Crit Care 24: 256
136. Guerin C, Reignier J, Richard JC, Beuret P, Gacouin A, Boulain T, Mercier E, Badet M, Mercat A, Baudin O, Clavel M, Chatellier D, Jaber S, Rosselli S, Mancebo J, Sirodot M, Hilbert G, Bengler C, Richecoeur J, Gainnier M, Bayle F, Bourdin G, Leray V, Girard R, Baboi L, Ayzac L, Group PS. 2013. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 368: 2159-68
137. Niu J, Straubinger RM, Mager DE. 2019. Pharmacodynamic Drug-Drug Interactions. Clin Pharmacol Ther 105: 1395-406
138. Palleria C, Di Paolo A, Giofrè C, Caglioti C, Leuzzi G, Siniscalchi A, De Sarro G, Gallelli L. 2013. Pharmacokinetic drug-drug interaction and their implication in clinical management. J Res Med Sci 18: 601-10
139. Tannenbaum C, Sheehan NL. 2014. Understanding and preventing drug-drug and drug-gene interactions. Expert Rev Clin Pharmacol 7: 533-44
140. Liverpool TUo. 2020. Liverpool COVID-19 Interactions. The University of Liverpool
141. Barlow A, Landolf KM, Barlow B, Yeung SYA, Heavner JJ, Claassen CW, Heavner MS. 2020. Review of Emerging Pharmacotherapy for the Treatment of Coronavirus Disease 2019. Pharmacotherapy 40: 416-37
142. Yang K. 2020. What Do We Know About Remdesivir Drug Interactions? Clin Transl Sci
143. Bobrowski T, Chen L, Eastman RT, Itkin Z, Shinn P, Chen C, Guo H, Zheng W, Michael S, Simeonov A, Hall MD, Zakharov AV, Muratov EN. 2020. Discovery of Synergistic and Antagonistic Drug Combinations against SARS-CoV-2 In Vitro. bioRxiv
144. Deng P, Zhong D, Yu K, Zhang Y, Wang T, Chen X. 2013. Pharmacokinetics, metabolism, and excretion of the antiviral drug arbidol in humans. Antimicrob Agents Chemother 57: 1743-55
145. Bishara D, Kalafatis C, Taylor D. 2020. Emerging and experimental treatments for COVID-19 and drug interactions with psychotropic agents. Ther Adv Psychopharmacol 10: 2045125320935306
146. Du YX, Chen XP. 2020. Favipiravir: Pharmacokinetics and Concerns About Clinical Trials for 2019-nCoV Infection. Clin Pharmacol Ther 108: 242-7
147. Zhao Y, Harmatz JS, Epstein CR, Nakagawa Y, Kurosaki C, Nakamura T, Kadota T, Giesing D, Court MH, Greenblatt DJ. 2015. Favipiravir inhibits acetaminophen sulfate formation but minimally affects systemic pharmacokinetics of acetaminophen. Br J Clin Pharmacol 80: 1076-85
148. Triggle CR, Bansal D, Farag E, Ding H, Sultan AA. 2020. COVID-19: Learning from Lessons To Guide Treatment and Prevention Interventions. mSphere 5
Widget de imagen
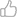 0
0
 0
0